Advanced Photochemical Reduction of Carbonyl to Methylene for Scalable Fine Chemical Production
The landscape of organic synthesis is undergoing a transformative shift towards sustainable methodologies, exemplified by the groundbreaking technology disclosed in patent CN113443950A. This intellectual property introduces a novel method for reducing carbonyl groups directly into methylene units under visible light illumination, specifically within the 380-456nm wavelength range. For R&D directors and process chemists seeking robust alternatives to classical reduction strategies, this patent offers a compelling solution that merges high atom utilization with exceptional safety profiles. The core innovation lies in the elimination of transition metal catalysts and harsh reagents, replacing them with readily available amine compounds and photon energy to drive the transformation. This approach not only streamlines the synthetic route by removing the need for additional protective group manipulations but also ensures that the final product residue toxicity is minimized to negligible levels. By leveraging this photochemical protocol, manufacturers can achieve a level of process control and environmental compliance that is increasingly demanded by global regulatory bodies in the pharmaceutical and agrochemical sectors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial reduction of carbonyl functionalities to methylene bridges has been dominated by two primary methodologies, both of which carry significant operational and environmental burdens that modern supply chains strive to eliminate. The first, known as the Clemmensen reduction, necessitates the reflux of aldehydes or ketones with zinc amalgam in concentrated hydrochloric acid, a process that is strictly limited to acid-stable compounds and generates substantial quantities of heavy metal waste requiring complex disposal procedures. The second conventional pathway, the Wolff-Kishner-Huang Minlon reduction, involves reacting carbonyls with hydrazine and potassium hydroxide in high-boiling solvents like diethylene glycol under intense heating, which poses severe safety risks due to the explosive nature of hydrazine and the energy-intensive conditions required. Furthermore, recent attempts to utilize transition metal catalysts such as Palladium or Nickel have introduced new challenges, including low catalytic activity, the necessity for expensive ligands, and the critical issue of residual metal contamination in the final active pharmaceutical ingredients (APIs). These traditional methods collectively represent a bottleneck in cost reduction in fine chemical manufacturing, as they demand rigorous purification steps to meet stringent purity specifications regarding heavy metals and toxic by-products.
The Novel Approach
In stark contrast to these legacy techniques, the method described in CN113443950A utilizes a metal-free, photo-driven mechanism that fundamentally alters the economic and safety dynamics of carbonyl reduction. By employing simple amine compounds, such as triethylamine or diisopropylethylamine, as the stoichiometric reductant in conjunction with visible light irradiation, the reaction proceeds under mild temperatures ranging from 20°C to 80°C. This gentle thermal profile prevents the degradation of sensitive functional groups that would otherwise decompose under the harsh acidic or basic conditions of classical reductions. The reaction system is characterized by its simplicity, requiring only the mixing of the carbonyl substrate and the amine in a common organic solvent like acetonitrile, followed by exposure to a 390nm LED light source for a duration of 12 to 24 hours. This eliminates the need for high-pressure reactors or specialized corrosion-resistant equipment, thereby drastically simplifying the operation in the preparation production process and lowering the capital expenditure required for plant infrastructure. Moreover, the absence of heavy metals means that the downstream purification process is significantly abbreviated, as there is no need for expensive scavenging resins or complex extraction protocols to remove trace catalyst residues.
Mechanistic Insights into Photochemical Carbonyl Reduction
The mechanistic underpinning of this transformation relies on the excitation of the carbonyl-amine complex or the carbonyl species itself upon absorption of photons in the near-UV to visible spectrum. When irradiated at wavelengths between 380nm and 456nm, the system enters an excited state that facilitates electron transfer from the lone pair of the nitrogen atom in the amine to the antibonding orbital of the carbonyl group. This single-electron transfer (SET) process generates a radical ion pair, which subsequently undergoes proton transfer and further reduction steps to cleave the carbon-oxygen double bond and form the stable carbon-hydrogen bonds of the methylene group. The use of amines like triethylamine is particularly effective because they serve dual roles as both the electron donor and the proton source, ensuring high atom economy throughout the reaction cycle. Unlike transition metal catalysis where the metal center cycles through various oxidation states, this organic photocatalysis or direct photolysis pathway avoids the introduction of any inorganic contaminants, ensuring that the impurity profile of the final product is composed primarily of organic by-products that are easier to separate via standard chromatographic techniques. This mechanistic clarity allows process chemists to predict substrate compatibility with greater accuracy, particularly for complex polycyclic structures where metal coordination might lead to unwanted side reactions or catalyst poisoning.
Furthermore, the control over impurities is inherently superior in this photochemical system due to the specificity of the light-induced activation. In traditional thermal reductions, high temperatures can promote non-selective bond cleavages or rearrangements, leading to a complex mixture of by-products that are difficult to resolve. However, the photon energy provided by the 390nm LED source is tuned to activate specific electronic transitions within the reactant molecules, thereby minimizing off-target reactivity. The patent data indicates that even substrates with diverse substituents, including halogens, alkoxy groups, and trifluoromethyl moieties, are tolerated without dehalogenation or demethylation, which are common failure modes in metal-catalyzed hydrogenolysis. This selectivity is crucial for maintaining the integrity of advanced intermediates used in the synthesis of high-value drugs and agrochemicals. Additionally, the reaction kinetics can be finely tuned by adjusting the light intensity and the molar ratio of the amine, typically kept between 1:3 and 1:6, allowing for optimization of the reaction rate without compromising the yield or purity. The result is a highly reproducible process that delivers consistent quality across different batches, a key requirement for reliable pharmaceutical intermediate supplier operations.
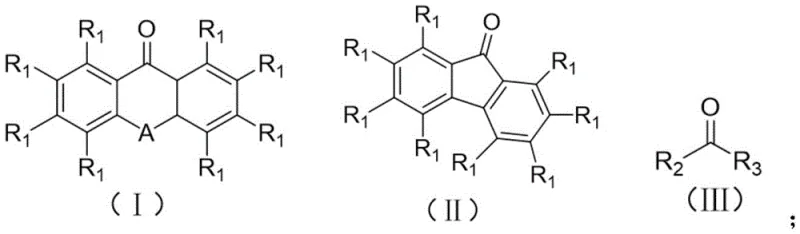
How to Synthesize 9H-Fluorene Derivatives Efficiently
To implement this technology for the synthesis of valuable scaffolds like 9H-fluorene and its derivatives, operators must adhere to precise stoichiometric and environmental controls to maximize yield and efficiency. The process begins with the dissolution of the specific carbonyl precursor, such as 9H-fluorenone, in an ultra-dry solvent like acetonitrile to prevent quenching of the excited states by moisture. Following the addition of the amine reductant, the mixture is subjected to continuous stirring under the specified LED illumination, ensuring uniform photon exposure throughout the reaction volume. While the general principles are straightforward, the detailed standardized synthesis steps see the guide below for specific parameters regarding temperature ramping and workup procedures tailored to different substrate classes.
- Mix the carbonyl substrate and amine compound (e.g., triethylamine) in a molar ratio of 1: (3-6) within a suitable organic solvent such as acetonitrile.
- Seal the reaction vessel and stir the mixture at temperatures between 20°C and 80°C while irradiating with a 380-456nm light source, preferably a 390nm LED.
- After 12-24 hours of reaction, remove the solvent via spin drying and purify the resulting methylene product using standard column chromatography techniques.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photochemical reduction technology translates into tangible strategic advantages that extend far beyond the laboratory bench. The primary value driver is the drastic simplification of the supply chain for raw materials, as the method utilizes commodity chemicals like triethylamine and acetonitrile which are globally available and price-stable, unlike specialized transition metal catalysts that are subject to geopolitical supply risks and volatile pricing. By eliminating the dependency on precious metals such as Palladium or Platinum, manufacturers can insulate their production costs from the fluctuations of the commodities market, ensuring more predictable budgeting and long-term cost stability. Furthermore, the reduction in process steps—specifically the removal of catalyst loading and subsequent metal scavenging—leads to a shorter overall cycle time, which enhances the responsiveness of the supply chain to sudden changes in market demand. This agility is critical in the fast-paced pharmaceutical industry where time-to-market for new drug candidates is a key competitive differentiator.
- Cost Reduction in Manufacturing: The economic impact of switching to this metal-free protocol is profound, primarily driven by the complete removal of expensive catalyst procurement and the associated waste disposal costs. Traditional methods often require catalyst loadings that, while small in percentage, represent a significant line item in the bill of materials when scaled to multi-ton production, not to mention the cost of licensed scavengers to reduce metal residues to ppm levels. By utilizing inexpensive amines and electricity for light generation, the variable cost per kilogram of the product is significantly lowered, allowing for more competitive pricing strategies in the global marketplace. Additionally, the mild reaction conditions reduce energy consumption related to heating and cooling, contributing to lower utility bills and a smaller carbon footprint, which aligns with corporate sustainability goals and may qualify for green manufacturing incentives.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures a more reliable supply of critical intermediates by mitigating the risks associated with complex chemical logistics. Since the reagents are non-hazardous and stable under ambient conditions, storage and transportation requirements are less stringent compared to pyrophoric reagents or high-pressure hydrogen gas used in catalytic hydrogenation. This ease of handling reduces the likelihood of shipping delays caused by hazardous material regulations and simplifies the inventory management process at manufacturing sites. Moreover, the scalability of LED photoreactor technology has matured significantly, meaning that scaling from pilot plant to commercial production does not require a complete redesign of the reaction engineering, thus ensuring continuity of supply during capacity expansions.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method offers a clear path to compliant and scalable manufacturing. The absence of heavy metals in the waste stream simplifies effluent treatment, reducing the load on wastewater treatment plants and minimizing the risk of regulatory fines related to discharge limits. The low toxicity of the reaction residues also improves workplace safety for operators, reducing the need for extensive personal protective equipment and specialized containment facilities. As global regulations on chemical manufacturing become increasingly stringent, adopting such green chemistry principles future-proofs the production facility against evolving compliance standards, ensuring uninterrupted operations and maintaining the social license to operate in sensitive industrial zones.
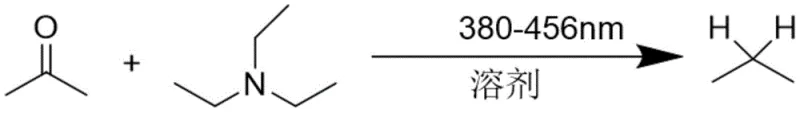
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photochemical reduction technology, derived directly from the experimental data and claims within the patent documentation. These insights are intended to clarify the operational feasibility and scope of application for potential partners evaluating this methodology for their own production pipelines. Understanding these nuances is essential for making informed decisions about process integration and technology transfer.
Q: What are the primary advantages of this photochemical reduction over Clemmensen reduction?
A: Unlike Clemmensen reduction which requires toxic zinc amalgam and concentrated hydrochloric acid, this photochemical method operates under mild, metal-free conditions, significantly reducing environmental pollution and simplifying waste treatment protocols.
Q: Is this method suitable for acid-labile substrates?
A: Yes, because the reaction does not rely on strong acidic or basic conditions like traditional Wolff-Kishner or Clemmensen methods, it is highly compatible with acid-labile and base-sensitive functional groups often found in complex pharmaceutical intermediates.
Q: What light source is required for industrial scale-up?
A: The patent specifies the use of blue LED light sources, particularly around 390nm (Kessil lamps), which are energy-efficient and easily scalable for flow chemistry or large batch photoreactors compared to high-energy UV sources.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9H-Fluorene Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the photochemical reduction technology detailed in CN113443950A and have integrated similar green chemistry principles into our own CDMO service offerings. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped to analyze and verify the absence of heavy metal contaminants, guaranteeing that every batch of high-purity pharmaceutical intermediate meets the exacting standards required by top-tier global clients. Our commitment to technological excellence allows us to offer customized synthesis solutions that leverage the latest advancements in photochemistry to deliver superior value.
We invite you to engage with our technical procurement team to discuss how this innovative reduction method can be applied to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this metal-free process for your target molecules. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a trusted partner in your supply chain for complex organic intermediates.
