Advanced Synthesis of Montelukast Sodium Intermediates for Commercial Scale-Up
Advanced Synthesis of Montelukast Sodium Intermediates for Commercial Scale-Up
The pharmaceutical industry constantly seeks more efficient pathways for producing high-volume active pharmaceutical ingredients (APIs), and the synthesis of Montelukast Sodium remains a critical focus area due to its widespread use in asthma management. Patent CN102424673B introduces a groundbreaking technology for synthesizing Montelukast Sodium and its novel intermediates, addressing long-standing challenges in cost and operational complexity. This patent details a method that utilizes cheap and easily accessible raw materials, resulting in a significantly shorter synthesis route that is ideally suited for industrial production. By shifting away from traditional reliance on precious metal catalysts, this innovation offers a compelling value proposition for manufacturers aiming to optimize their production lines while maintaining stringent quality standards.
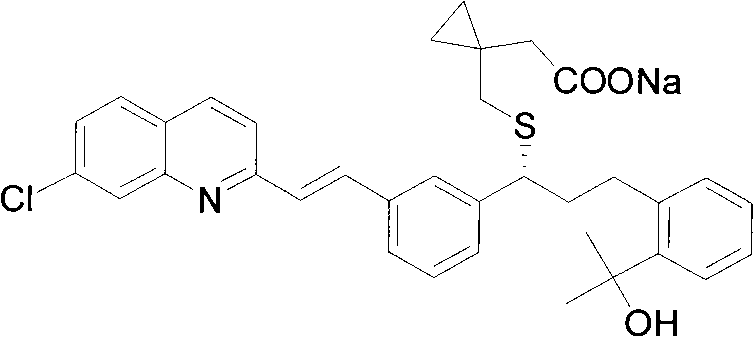
For procurement managers and supply chain directors, the implications of this technology are profound, as it promises to stabilize the supply of reliable pharmaceutical intermediates supplier networks. The core of this invention lies in the preparation of a specific intermediate, designated as Formula I in the patent documentation, which serves as a versatile building block for the final API. The methodology described allows for the efficient construction of the complex carbon skeleton required for Montelukast, bypassing several inefficient steps found in legacy processes. This strategic shift not only enhances the economic viability of the production process but also aligns with modern green chemistry principles by reducing the environmental footprint associated with heavy metal usage.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Montelukast Sodium has relied heavily on methodologies that present significant economic and operational hurdles for large-scale manufacturers. Prior art, such as the methods described in early patents by major pharmaceutical companies, often necessitates the use of valuable metallic palladium reagents for coupling reactions, which drastically inflates the cost of goods sold. Furthermore, many conventional routes require repeated Grignard reactions under harsh conditions that demand strict control of temperature and moisture, leading to higher rates of byproduct formation and lower overall yields. These complex sequences often result in intermediates that are unstable and unsuitable for long-term storage, creating bottlenecks in the supply chain and complicating inventory management for global producers.
The Novel Approach
In stark contrast, the novel approach outlined in CN102424673B leverages a base-catalyzed alkylation strategy inspired by green chemistry advancements, specifically utilizing potassium hydroxide or sodium hydroxide to facilitate key bond formations. This method avoids the need for severe toxicity and expensive heavy metal catalysts, thereby simplifying the downstream purification processes and eliminating the risk of metal contamination in the final product. The new route is characterized by its operational simplicity and the use of robust starting materials that are commercially available in bulk quantities, ensuring a steady flow of production. By streamlining the synthesis into fewer steps with higher atom economy, this approach effectively reduces the lead time for high-purity pharmaceutical intermediates and enhances the overall sustainability of the manufacturing process.
Mechanistic Insights into Base-Catalyzed Alkylation and Protection Strategies
The chemical ingenuity of this patent is best understood through the detailed mechanistic steps involved in constructing the key intermediate. The process begins with the reaction of phthalide (Formula IV) with a Grignard reagent, specifically methyl magnesium halide, to form a diol intermediate (Formula V). This step is critical as it establishes the gem-dimethyl motif adjacent to the aromatic ring, which is essential for the biological activity of the final drug. Following this, a selective protection strategy is employed where the primary hydroxyl group is blocked using protecting groups such as acetyl or silyl groups, while the tertiary hydroxyl is subsequently protected, often with a tetrahydropyranyl (THP) group. This orthogonal protection scheme allows for precise chemical manipulation in subsequent steps without affecting sensitive functional groups.
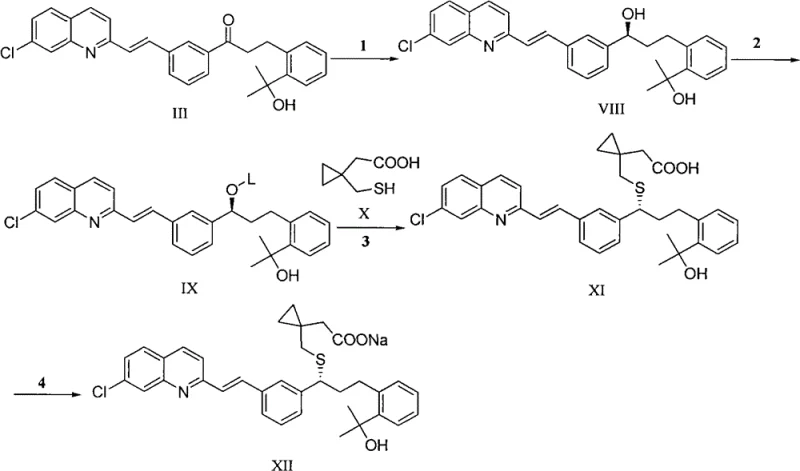
Following the construction of the protected intermediate, the synthesis proceeds through a pivotal coupling reaction where the intermediate reacts with a quinoline derivative under basic conditions. This alkylation step forms the carbon-carbon bond linking the two major aromatic systems of the molecule. The resulting mixture is then subjected to selective oxidation to generate the ketone functionality, followed by deprotection to reveal the necessary hydroxyl groups for the final transformation. The final stages involve a chiral reduction to establish the correct stereochemistry at the secondary alcohol position, followed by nucleophilic substitution with a cyclopropane-containing thiol to install the sulfur linkage. This sequence demonstrates a high level of control over regioselectivity and stereoselectivity, ensuring that the final Montelukast Sodium meets the rigorous purity specifications required for pharmaceutical applications.
How to Synthesize Montelukast Sodium Efficiently
The synthesis of Montelukast Sodium via this novel route involves a series of well-defined chemical transformations that prioritize yield and purity. The process initiates with the preparation of the protected phenyl-ethanol intermediate, followed by its coupling with the quinoline vinyl component to form the ketone precursor. Subsequent steps involve chiral reduction to set the stereocenter, activation of the alcohol as a leaving group, and finally, displacement with the cyclopropane thiol acid to form the free acid, which is then converted to the sodium salt. For a comprehensive breakdown of the specific reaction conditions, stoichiometry, and workup procedures required to execute this synthesis in a GMP environment, please refer to the detailed technical guide below.
- Synthesize the key protected intermediate (Formula I) starting from phthalide via Grignard reaction, followed by selective hydroxyl protection and deprotection steps.
- Perform base-catalyzed alkylation between the protected intermediate and quinoline derivatives to form the ketone precursor (Formula III).
- Convert the ketone intermediate to Montelukast Sodium through chiral reduction, thioether formation, and final salt formation.
Commercial Advantages for Procurement and Supply Chain Teams
For decision-makers focused on the bottom line and operational continuity, the adoption of this synthesis route offers substantial strategic benefits that extend beyond mere technical feasibility. The elimination of palladium catalysts represents a direct reduction in raw material expenditure, as precious metals constitute a significant portion of the variable costs in traditional API manufacturing. Additionally, the simplified workflow reduces the requirement for specialized equipment capable of handling pyrophoric reagents or maintaining ultra-low temperatures, thereby lowering capital expenditure and maintenance overheads. This efficiency translates into a more competitive pricing structure for the final intermediate, allowing downstream partners to achieve better margins in a highly competitive generic pharmaceutical market.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts and the use of common bases like potassium hydroxide drastically lowers the cost of reagents per kilogram of product. Furthermore, the simplified purification steps reduce solvent consumption and waste disposal costs, contributing to a leaner and more cost-effective production model that maximizes resource utilization.
- Enhanced Supply Chain Reliability: By relying on commodity chemicals rather than specialized organometallic reagents, manufacturers can mitigate the risk of supply disruptions caused by geopolitical issues or vendor shortages. The robustness of the intermediates produced allows for longer storage times and easier transportation, facilitating a more flexible and resilient global distribution network for critical pharmaceutical ingredients.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of heavy metals make this process inherently safer and easier to scale from pilot batches to multi-ton commercial production. This alignment with environmental regulations reduces the regulatory burden associated with heavy metal clearance testing and waste treatment, ensuring smoother audits and faster time-to-market for new generic filings.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is crucial for stakeholders evaluating its potential impact on their production portfolios. The following questions address common inquiries regarding the practical implementation and benefits of this patented technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for technical assessment.
Q: What are the primary advantages of this synthesis route over conventional palladium-catalyzed methods?
A: This novel route eliminates the need for expensive and toxic palladium catalysts, significantly reducing raw material costs and simplifying the purification process by removing heavy metal residues.
Q: How does this method improve supply chain stability for Montelukast production?
A: By utilizing cheap and readily available starting materials like phthalide and avoiding complex multi-step Grignard sequences, the method ensures a more robust and continuous supply of high-purity intermediates.
Q: Is this synthesis method suitable for large-scale industrial manufacturing?
A: Yes, the process features simple operations, mild reaction conditions, and high yields, making it highly adaptable for commercial scale-up from pilot plants to multi-ton annual production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Montelukast Sodium Intermediate Supplier
As the global demand for respiratory medications continues to grow, securing a stable source of high-quality intermediates is paramount for pharmaceutical success. NINGBO INNO PHARMCHEM stands ready to support your production needs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of Montelukast Sodium intermediate we deliver adheres to the highest international standards. We understand the critical nature of API supply chains and are committed to providing consistent quality and reliability.
We invite you to collaborate with us to explore how this advanced synthesis route can be integrated into your manufacturing strategy to drive efficiency and reduce costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your long-term growth and operational excellence in the pharmaceutical sector.
