Advanced Oxidative Coupling Strategy for Scalable Production of Functionalized Phosphoramide Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex molecular architectures, particularly those containing sensitive heteroatom linkages. Patent CN110845534A introduces a groundbreaking preparation method for phosphoramide compounds that addresses long-standing challenges in organic synthesis. This technology leverages a sophisticated oxidative coupling reaction between a phosphoramidate precursor and an alkene derivative under an oxygen atmosphere. Unlike traditional approaches that often suffer from low yields or poor stability of the phosphorus-nitrogen bond, this novel process utilizes a synergistic dual-transition metal catalyst system. The result is the efficient generation of phosphoramide compounds containing unsaturated carbon-carbon double bonds with remarkable stereoselectivity. For R&D directors and procurement specialists, this represents a significant advancement in accessing high-value intermediates for drug discovery and material science applications, ensuring a reliable supply of complex building blocks.
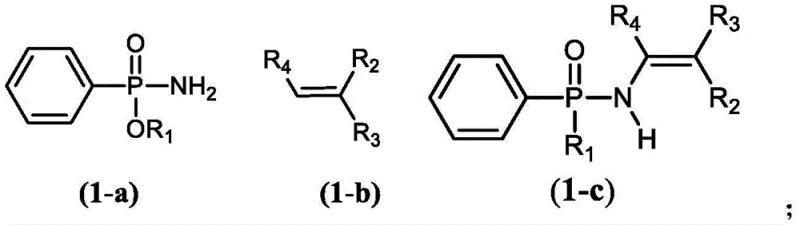
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of phosphoramides has been fraught with difficulties due to the inherent instability of the P-N bond compared to the more robust N-H bond. Conventional synthetic routes often require harsh reaction conditions, such as strong acids or bases, high temperatures, or stoichiometric amounts of hazardous reagents, which can lead to the decomposition of the phosphoramide core. Furthermore, achieving high stereoselectivity in the formation of adjacent double bonds has been a persistent hurdle, often resulting in complex mixtures of E and Z isomers that are difficult and costly to separate. These limitations not only reduce the overall process efficiency but also introduce significant impurities that complicate downstream purification, thereby increasing the cost of goods sold and extending the lead time for high-purity pharmaceutical intermediates. The lack of mild, catalytic methods has restricted the widespread application of phosphoramides in asymmetric synthesis and materials science.
The Novel Approach
The methodology disclosed in CN110845534A offers a transformative solution by employing a catalytic dehydrogenation coupling reaction under mild oxidative conditions. By utilizing a first transition metal catalyst, specifically palladium-based, in conjunction with a second transition metal salt such as copper, cobalt, iron, or nickel, the process facilitates the direct coupling of the phosphoramidate nitrogen with the alkene substrate. This approach operates effectively at moderate temperatures ranging from 50°C to 100°C, typically around 80°C, and uses molecular oxygen as the terminal oxidant, which is both economical and environmentally benign. The reaction demonstrates exceptional tolerance for various functional groups on both the phosphorus and alkene components, allowing for the synthesis of a diverse library of derivatives. This shift from stoichiometric, harsh conditions to a catalytic, aerobic process significantly enhances the feasibility of commercial scale-up of complex phosphoramide intermediates while maintaining high product integrity.
Mechanistic Insights into Pd-Cu Dual Catalytic Oxidative Coupling
The success of this synthesis relies on the intricate interplay between the palladium and copper catalysts within the reaction cycle. The proposed mechanism involves the coordination of the alkene to the palladium center, followed by nucleophilic attack by the phosphoramidate nitrogen. The presence of the second transition metal, particularly copper salts like CuBr or CuCl, plays a crucial role in facilitating the regeneration of the active palladium species and managing the electron transfer processes required for the oxidative coupling. Experimental evidence suggests that the reaction proceeds through a pathway that favors the formation of the Z-isomer, likely due to steric and electronic factors governed by the catalyst-ligand environment. The use of oxygen as the oxidant ensures that the catalytic cycle is closed efficiently without generating excessive waste. For technical teams, understanding this mechanism is vital for optimizing reaction parameters such as catalyst loading and solvent choice to maximize yield and selectivity.
Furthermore, the patent highlights the critical influence of chloride ions on the reaction outcome, particularly when targeting chlorinated derivatives. When the reaction system contains chloride ions, and the molar ratio of substrates to chloride is controlled precisely, the mechanism shifts towards a cross-coupling chlorination pathway. This involves a Wacker-type process where Pd-alkene coordination is followed by electrophilic chlorination, driven by the continuous generation of Cl+ species under oxygen atmosphere. This mechanistic flexibility allows chemists to toggle between producing simple unsaturated phosphoramides and more complex chlorinated variants simply by adjusting the chloride source and concentration. Such control over the reaction pathway is essential for impurity control, as it minimizes the formation of unwanted by-products and ensures that the final product meets stringent purity specifications required for pharmaceutical applications.
How to Synthesize Phosphoramide Compounds Efficiently
To implement this synthesis effectively, precise control over reaction conditions is paramount. The process begins with the selection of appropriate substrates, where the phosphoramidate (Formula 1-a) and the alkene (Formula 1-b) are chosen based on the desired final structure. The reaction is typically conducted in solvents such as tetrahydrofuran (THF), toluene, or xylene, with THF often providing optimal results. The catalyst system, comprising Pd(OAc)2 and CuBr, is added in specific molar ratios relative to the substrates to ensure complete conversion. The mixture is then heated under an oxygen atmosphere, usually at 1 atm pressure, for a duration of approximately 5 hours. Detailed standard operating procedures for scaling this reaction from gram to kilogram scale involve careful monitoring of oxygen uptake and temperature profiles to maintain safety and consistency. The detailed standardized synthesis steps are outlined in the guide below.
- Prepare the reaction mixture by combining the phosphoramidate precursor (Formula 1-a), the alkene substrate (Formula 1-b), and a suitable organic solvent such as tetrahydrofuran or toluene.
- Add the dual catalyst system consisting of a first transition metal catalyst (e.g., Pd(OAc)2) and a second transition metal catalyst (e.g., CuBr) to the reaction vessel under an oxygen atmosphere.
- Heat the reaction mixture to a temperature between 50°C and 100°C, preferably 80°C, and maintain stirring for approximately 5 hours to achieve high stereoselectivity and yield.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers substantial advantages that directly impact the bottom line and supply chain resilience. The shift towards a catalytic aerobic oxidation process eliminates the need for expensive and hazardous stoichiometric oxidants, which significantly reduces raw material costs and waste disposal expenses. The use of readily available starting materials, such as simple alkenes and phosphoramidates, ensures a stable supply chain that is less susceptible to market volatility compared to specialized reagents. Additionally, the high stereoselectivity of the reaction reduces the burden on downstream purification processes, leading to higher overall throughput and reduced production cycles. For procurement managers, this translates into a more predictable cost structure and the ability to secure high-purity intermediates at competitive prices, supporting cost reduction in pharmaceutical intermediate manufacturing.
- Cost Reduction in Manufacturing: The elimination of harsh reagents and the use of molecular oxygen as a green oxidant drastically simplify the workup procedure. By avoiding the generation of heavy metal waste associated with stoichiometric oxidants, the process lowers environmental compliance costs. The high yield and selectivity mean less raw material is wasted on off-spec products, leading to substantial cost savings in the overall production budget. Furthermore, the catalyst loading can be optimized to minimize the usage of precious palladium, further enhancing the economic viability of the process for large-scale operations.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like THF, oxygen, and common copper salts ensures that the supply chain is robust and less prone to disruptions. Unlike processes requiring exotic ligands or unstable intermediates, this method utilizes stable reagents that can be sourced from multiple vendors globally. This diversification of supply sources mitigates the risk of single-source dependency, ensuring continuous production capability. The mild reaction conditions also reduce the risk of safety incidents, contributing to a more reliable and uninterrupted manufacturing schedule for critical pharmaceutical ingredients.
- Scalability and Environmental Compliance: The process is inherently scalable due to its mild thermal requirements and the use of gas-liquid reactions that can be managed in standard reactor setups. The reduced generation of hazardous waste aligns with increasingly strict environmental regulations, making it easier to obtain necessary permits for expansion. The ability to produce high-purity products with minimal purification steps supports the rapid scale-up from pilot plant to commercial production volumes. This scalability ensures that the technology can meet growing market demand without compromising on quality or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this phosphoramide synthesis technology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation. Understanding these aspects helps stakeholders make informed decisions regarding process adoption and integration into existing manufacturing workflows. The answers reflect the proven capabilities of the method in handling diverse substrates and maintaining high performance metrics.
Q: How does this method address the stability issues of P-N bonds in phosphoramides?
A: Traditional methods often struggle with the fragility of P-N bonds during functionalization. This patented oxidative coupling approach utilizes a mild Pd-Cu catalytic system under oxygen atmosphere, which facilitates the formation of the C-N bond without degrading the sensitive phosphoramide core, ensuring structural integrity.
Q: What is the stereoselectivity profile of the resulting phosphoramide compounds?
A: The process demonstrates exceptional stereocontrol, particularly for the Z-isomer. Experimental data indicates that the molar ratio of Z/E configurations can exceed 20:1 in optimized conditions, which is critical for applications requiring specific geometric isomers in asymmetric synthesis.
Q: Can this synthesis route be adapted for chlorinated phosphoramide derivatives?
A: Yes, by adjusting the chloride ion concentration in the reaction system (specifically controlling the molar ratio of substrates to chloride ions to be less than or equal to 1), the method can be tuned to produce chlorinated phosphoramide compounds (Formula 1-d) via a cross-coupling chlorination mechanism.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphoramide Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to drive innovation in drug development. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex molecules like those described in CN110845534A can be manufactured reliably. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation. Whether you require custom synthesis of specific phosphoramide derivatives or large-scale supply of key intermediates, our infrastructure is designed to support your project from early-stage development through to commercial launch.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us to request specific COA data and route feasibility assessments for your target molecules. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable phosphoramide compound supplier dedicated to excellence, efficiency, and long-term partnership in the global pharmaceutical supply chain.
