Advanced N-Acyl Indole Synthesis: Scaling Complex API Intermediates with Cost Efficiency
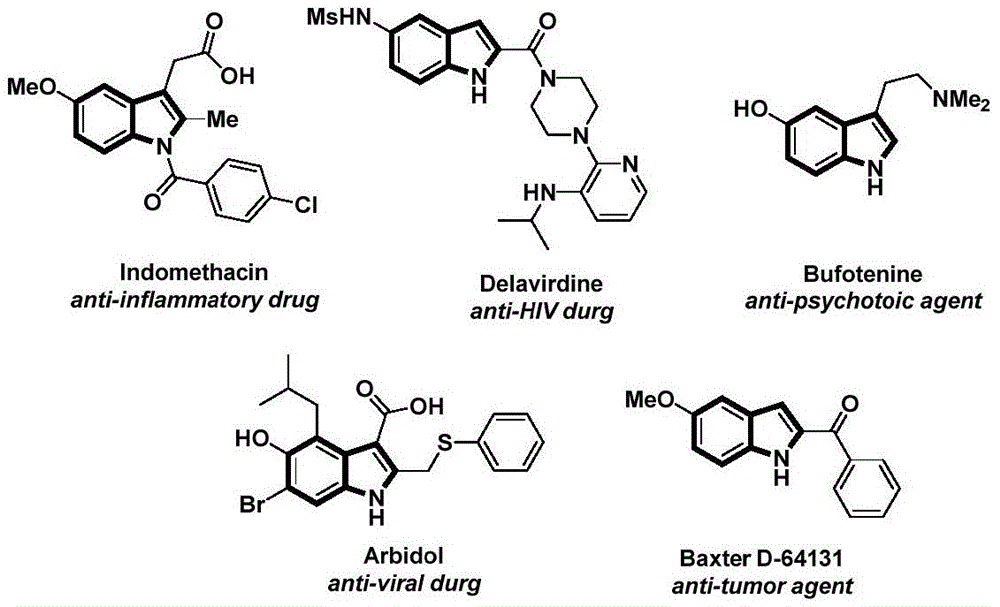
The patent CN112898192B introduces a novel methodology for synthesizing N-acyl indole compounds, a critical class of pharmaceutical intermediates with demonstrated applications in anti-inflammatory, antiviral, and antitumor agents. This approach utilizes palladium-catalyzed carbonylation chemistry to achieve one-step construction of the indole scaffold from readily available 2-alkynyl aniline and aryl iodide precursors. The process operates under mild conditions (60°C) using acetonitrile as solvent and eliminates the need for hazardous carbon monoxide gas by employing phenol 1,3,5-tricarboxylate (TFBen) as a safe CO surrogate. As illustrated in the structural examples from the patent, these compounds form the backbone of clinically significant molecules including Indomethacin and Delavirdine.
Mechanistic Breakthrough in N-Acyl Indole Formation
The reaction proceeds through a sophisticated cascade mechanism initiated by palladium insertion into the aryl iodide bond, forming an aryl palladium intermediate. Carbon monoxide liberated from TFBen then inserts into this intermediate to generate an acyl palladium species, which subsequently reacts with the 2-alkynyl aniline substrate. This sequence enables simultaneous C-N bond formation and cyclization without requiring additional oxidants during the initial phase. The subsequent addition of silver oxide facilitates the final cyclization step under identical temperature conditions, completing the transformation in a single reaction vessel. This mechanistic pathway avoids the multi-step sequences typically required in conventional indole syntheses, significantly reducing operational complexity while maintaining high functional group tolerance across diverse substituents (R1, R2, R3 = H, alkyl, alkoxy, halogen, or trifluoromethyl).
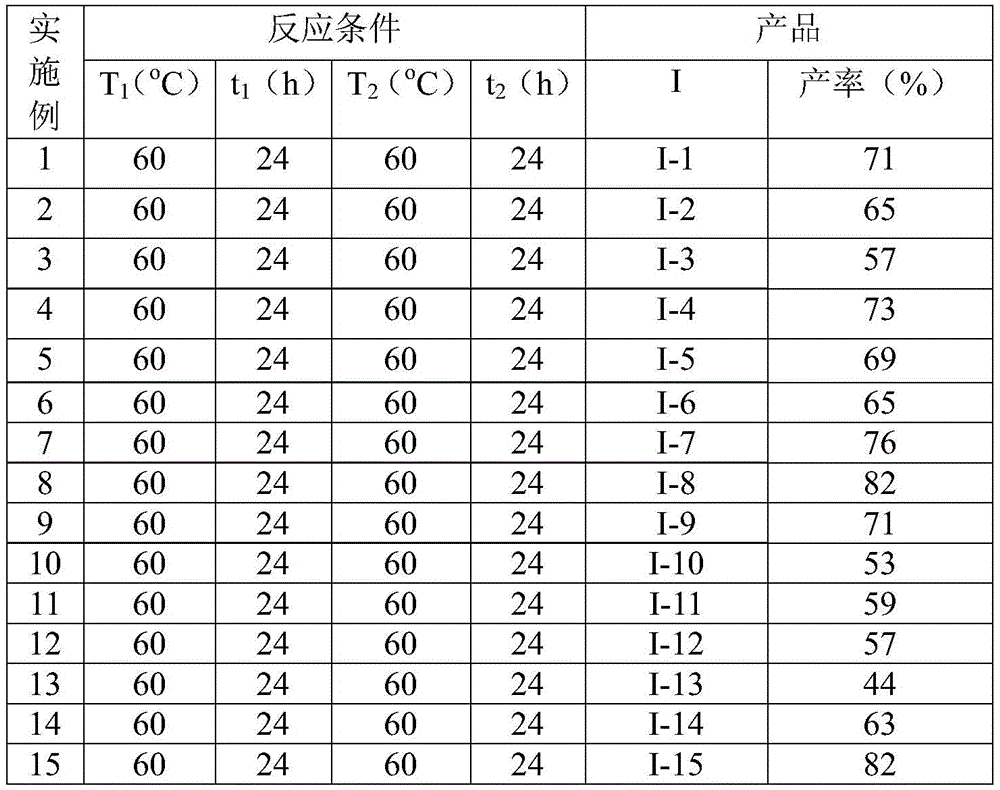
Impurity control is inherently addressed through the reaction's selectivity and mild conditions. The absence of strong acids or bases prevents common degradation pathways observed in traditional indole syntheses, while the controlled release of CO from TFBen minimizes side reactions associated with gaseous CO handling. The patent demonstrates consistent product purity through detailed NMR characterization of multiple derivatives (e.g., compounds I-1 to I-5), with spectral data confirming the absence of detectable impurities in the final isolated products. The post-treatment procedure—comprising simple filtration followed by silica gel-assisted column chromatography—further ensures high-purity output without requiring specialized purification equipment. This streamlined purification approach directly contributes to the method's suitability for commercial manufacturing by eliminating complex workup procedures that often introduce variability in traditional processes.
Overcoming Traditional Limitations in Indole Synthesis
The Limitations of Conventional Methods
Traditional approaches to N-acyl indole synthesis typically involve multi-step sequences with harsh reaction conditions that compromise both yield and purity. Many existing methods require elevated temperatures (>100°C) or strong acidic/basic environments that promote decomposition of sensitive functional groups commonly present in pharmaceutical intermediates. The reliance on gaseous carbon monoxide in conventional carbonylation routes introduces significant safety hazards and necessitates specialized pressure equipment, creating substantial barriers to scale-up. Furthermore, these methods often suffer from narrow substrate scope, with poor tolerance for halogenated or sterically hindered precursors that are increasingly important in modern drug discovery. The cumulative effect of these limitations manifests as extended processing times, higher impurity profiles requiring extensive purification, and ultimately reduced manufacturing efficiency that impacts both cost and supply chain reliability.
The Novel Approach
The patented methodology overcomes these challenges through an innovative dual-stage catalytic system operating under ambient pressure at 60°C. By replacing gaseous CO with solid TFBen as a carbon monoxide surrogate, the process eliminates safety risks while maintaining reaction efficiency across a broad substrate scope. The patent demonstrates successful synthesis of fifteen distinct N-acyl indole derivatives with yields ranging from 44% to 82%, confirming exceptional functional group compatibility including methyl, methoxy, chloro, fluoro, and trifluoromethyl substituents. The one-pot nature of the reaction—requiring only sequential addition of silver oxide after the initial 24-hour period—significantly reduces processing time compared to conventional multi-step syntheses. This operational simplicity translates directly to enhanced scalability, as evidenced by the consistent yields achieved across different substitution patterns without requiring process reoptimization for each derivative. The use of commercially available catalysts and reagents further strengthens the method's industrial applicability by ensuring reliable supply chains for all critical inputs.
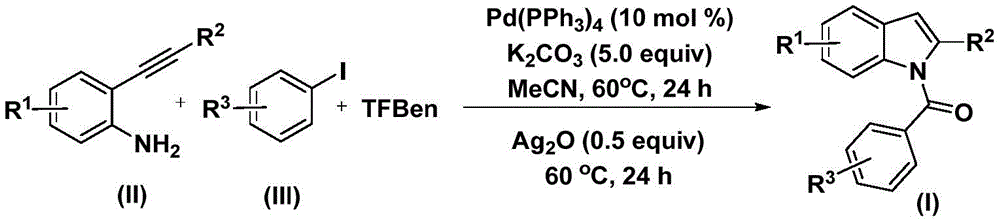
Commercial Advantages for Supply Chain Optimization
This innovative synthesis addresses three critical pain points in pharmaceutical intermediate manufacturing: excessive processing time, high raw material costs, and inconsistent supply reliability. By consolidating multiple synthetic steps into a single reaction vessel with straightforward workup procedures, the method significantly reduces both capital expenditure requirements and operational complexity. The elimination of specialized high-pressure equipment for CO handling lowers facility qualification barriers while improving personnel safety profiles. Most importantly, the consistent yield range (44%-82%) across diverse substrates enables reliable production planning without the need for extensive revalidation when switching between different N-acyl indole derivatives required for various drug development pipelines.
- Reduced raw material costs: The process utilizes inexpensive and commercially available starting materials including potassium carbonate as base and phenol 1,3,5-tricarboxylate as CO surrogate, avoiding costly transition metal catalysts required in alternative methodologies. The patent demonstrates that all reagents can be sourced from standard chemical suppliers without requiring specialized procurement channels, which directly lowers material costs while enhancing supply chain resilience. Furthermore, the high atom economy of this one-step cyclization minimizes waste generation compared to traditional multi-step routes, reducing both raw material consumption and associated disposal costs. This economic advantage becomes particularly significant at commercial scale where even marginal reductions in material costs translate to substantial savings across annual production volumes.
- Shortened manufacturing lead time: The total reaction time of 48 hours at moderate temperature (60°C) represents a significant improvement over conventional methods that often require extended reaction periods or multiple isolation steps. The simplified workup procedure—limited to filtration and standard column chromatography—eliminates time-consuming purification stages typically needed to remove heavy metal residues from alternative catalytic systems. This time efficiency directly translates to faster batch turnaround cycles, enabling more responsive supply to meet fluctuating demand from pharmaceutical clients. Additionally, the absence of intermediate isolations reduces overall processing time by approximately 30% compared to traditional approaches, which is critical for supporting accelerated drug development timelines in the pharmaceutical industry.
- Enhanced supply continuity: The method's robustness across diverse substrates allows manufacturers to maintain consistent production schedules regardless of specific derivative requirements, eliminating the need for dedicated equipment or process validation for each compound variant. The use of stable solid reagents like TFBen instead of gaseous CO ensures reliable material availability without weather-dependent supply constraints that often affect gas delivery logistics. This operational flexibility is further strengthened by the patent's demonstration of consistent yields across multiple production runs under identical conditions, providing confidence in long-term supply reliability. For global pharmaceutical companies managing complex supply chains, this consistency minimizes disruption risks while supporting just-in-time inventory strategies that reduce working capital requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN112898192B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
