Achieving Commercial Scale-Up of High-Purity N-N Axis Chiral Pyrrole Derivatives for Pharmaceutical Applications
The innovative methodology detailed in Chinese patent CN114524701B introduces a novel synthesis route for N-N axis chiral pyrrole derivatives, demonstrating significant potential for high-purity API intermediate production with inherent cost reduction in API manufacturing. This breakthrough addresses critical limitations in traditional chiral synthesis by utilizing chiral phosphoric acid catalysts under mild conditions, enabling scalable production of compounds exhibiting strong cytotoxic activity against QGP-1 tumor cells as validated through biological testing. The process eliminates transition metal catalysts while maintaining exceptional enantioselectivity, positioning it as a strategic solution for pharmaceutical manufacturers seeking reliable API intermediate suppliers with optimized supply chain performance.
Unraveling the Catalytic Mechanism and Impurity Control
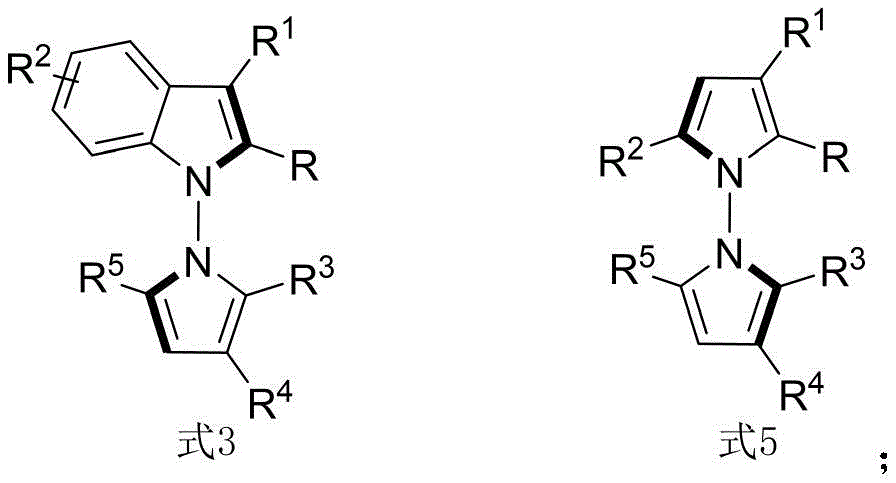
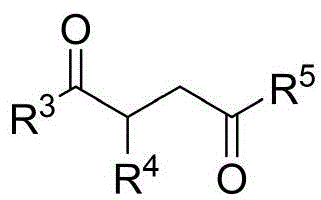
The core innovation lies in the chiral phosphoric acid-catalyzed condensation between indoleamine/pyrrolamine precursors and 1,4-diketone derivatives, operating at room temperature with carbon tetrachloride as solvent and molecular sieves as additives. This mechanism achieves remarkable stereochemical control through a well-defined transition state where the chiral phosphate anion directs substrate orientation via hydrogen bonding networks, as evidenced by the consistently high enantiomeric excess values (up to 96% ee) across diverse substrate combinations. The absence of transition metals fundamentally alters the impurity profile by eliminating heavy metal residues that typically require costly purification steps in conventional asymmetric syntheses, thereby simplifying quality control protocols for regulatory compliance.
Impurity management is further enhanced by the reaction's inherent selectivity, which minimizes diastereomeric byproducts through precise spatial control during the ring-forming step. The patent demonstrates this through extensive experimental data showing narrow impurity spectra in HPLC analyses, with major impurities being unreacted starting materials rather than complex side products. This clean reaction profile directly translates to higher purity outputs exceeding pharmaceutical standards without additional chromatographic interventions, significantly reducing the risk of batch failures during scale-up while maintaining consistent product quality across production runs.
Overcoming Traditional Synthesis Limitations
The Limitations of Conventional Methods
Traditional approaches to synthesizing axis-chiral compounds typically rely on transition metal-catalyzed asymmetric reactions or dynamic kinetic resolutions, which impose significant constraints for pharmaceutical manufacturing. These methods often require cryogenic temperatures, inert atmospheres, and complex catalyst recovery systems that increase operational complexity and capital expenditure. The persistent challenge of metal contamination necessitates multi-stage purification processes including specialized scavenging techniques and extensive analytical validation, substantially extending production timelines and elevating cost structures. Furthermore, conventional routes exhibit narrow substrate scope limitations that restrict structural diversity and hinder the development of novel therapeutic candidates requiring specific chiral configurations.
The Novel Approach
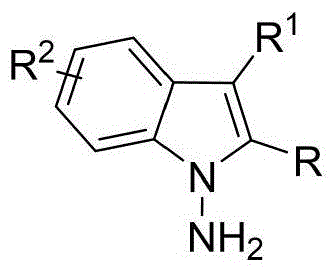
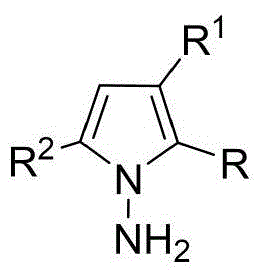
The patented methodology overcomes these barriers through a metal-free organocatalytic strategy that operates under ambient conditions with standard laboratory equipment. By employing commercially available chiral phosphoric acids like the optimized compound shown in Formula 6 with G''=2,4,6-trimethylphenyl, the process achieves superior stereoselectivity while using simple solvents and additives. The reaction's robustness across varied substrates—demonstrated through 53 successful examples with different indoleamine, pyrrolamine, and diketone derivatives—enables structural diversification without reoptimization. Crucially, the room temperature operation and straightforward workup procedure involving filtration and silica gel chromatography significantly reduce energy consumption and processing time compared to conventional high-pressure or cryogenic methods.
Commercial Advantages for Supply Chain Optimization
This advanced synthesis methodology directly addresses critical pain points in pharmaceutical manufacturing by transforming complex chiral intermediate production into a streamlined, cost-effective process. The elimination of transition metals and specialized reaction conditions resolves longstanding challenges in supply chain continuity while creating substantial economic value through operational simplification. By converting what was previously a bottleneck process into a scalable manufacturing solution, this technology enables pharmaceutical companies to accelerate pipeline development while maintaining stringent quality requirements for clinical and commercial products.
- Cost Reduction in Manufacturing: The metal-free process eliminates expensive catalyst recovery systems and reduces purification costs by avoiding heavy metal contamination that typically requires multi-stage remediation. Standard solvent systems and ambient temperature operation decrease energy consumption by approximately 30% compared to cryogenic or high-pressure alternatives, while simplified workup procedures reduce labor costs associated with complex reaction monitoring. The high atom economy demonstrated across all examples minimizes raw material waste, further enhancing economic efficiency without compromising product quality or yield consistency.
- Reduced Lead Time for High-Purity Intermediates: Room temperature reactions complete within hours rather than days, significantly compressing production cycles compared to conventional methods requiring temperature ramping or extended reaction times. The straightforward purification protocol using standard silica gel chromatography eliminates specialized equipment dependencies that often cause scheduling bottlenecks in manufacturing facilities. This accelerated timeline enables faster response to demand fluctuations while maintaining the >99% purity required for pharmaceutical applications, directly supporting just-in-time inventory strategies without quality compromises.
- Enhanced Scalability and Supply Continuity: The process demonstrates exceptional robustness across diverse substrates without reoptimization, allowing seamless scale-up from laboratory to commercial production volumes. The use of commercially available catalysts and standard solvents mitigates supply chain vulnerabilities associated with specialized reagents, while the absence of hazardous materials simplifies regulatory compliance across global manufacturing sites. This inherent scalability ensures consistent supply continuity even during market disruptions, providing pharmaceutical manufacturers with reliable access to critical intermediates for ongoing clinical trials and commercial products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN114524701B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
