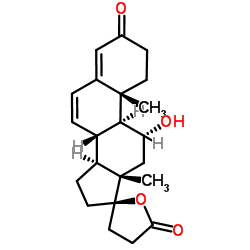
11α-Hydroxy Canrenone
- CAS No.192569-17-8
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity 11α-Hydroxy Canrenone (CAS 192569-17-8) designed for pharmaceutical synthesis. Reliable supply for Eplerenone production with comprehensive COA support.
Request Bulk PricingProduct Technical Details
Product Overview
11α-Hydroxy Canrenone represents a critical advanced intermediate within the steroid chemical family, specifically engineered for complex pharmaceutical synthesis pathways. As a key derivative in the development of mineralocorticoid receptor antagonists, this compound serves as an essential building block for modern cardiovascular therapeutic agents. Our manufacturing process ensures exceptional stereochemical integrity at the 11-alpha position, which is vital for the biological activity of downstream active pharmaceutical ingredients. We prioritize rigorous quality control measures to deliver material that meets the demanding standards of global pharmaceutical research and production facilities.
The chemical structure features a specialized oxidation state that facilitates further functionalization during multi-step synthesis routes. Maintaining high purity levels is paramount to prevent side reactions and ensure optimal yield in subsequent manufacturing stages. Our facility employs state-of-the-art analytical instrumentation to verify molecular identity and assay content before every shipment. This commitment to quality supports our partners in achieving regulatory compliance and efficient scale-up processes for their final drug products.
Technical Specifications
We adhere to strict internal quality standards that often exceed industry norms for pharmaceutical intermediates. Each batch undergoes comprehensive testing including high-performance liquid chromatography and nuclear magnetic resonance spectroscopy. The following table outlines the typical physical and chemical properties associated with this grade of material.
| Parameter | Specification |
|---|---|
| CAS Number | 192569-17-8 |
| Molecular Formula | C22H28O4 |
| Molecular Weight | 356.455 g/mol |
| Appearance | Yellow crystalline powder |
| Assay | ≥98.0% |
| Melting Point | 232-234°C |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 584.7±50.0 °C at 760 mmHg |
| Flash Point | 207.3±23.6 °C |
Industrial Applications
The primary application of 11α-Hydroxy Canrenone lies in the synthesis of Eplerenone, a selective aldosterone blocker used in the management of hypertension and heart failure. As a precursor, its quality directly influences the purity profile of the final active ingredient. Pharmaceutical manufacturers rely on consistent intermediate quality to streamline their validation processes and reduce production risks. Our material is suitable for both process development laboratories and large-scale commercial manufacturing environments.
Beyond its primary use, this compound serves as a valuable reference standard for analytical method development within quality control departments. Researchers utilize it to establish calibration curves and validate purification protocols. The stability of the hydroxy group under various reaction conditions makes it a versatile component in organic synthesis strategies focused on steroidal backbones. We support our clients with technical data packages that facilitate smooth technology transfer and regulatory filings.
Quality Assurance and Storage
Proper handling and storage are essential to maintain the integrity of this chemical intermediate. We recommend storing the material in a cool, ventilated environment away from direct sunlight and moisture sources. Our standard packaging consists of 25 kg drums, which are designed to protect the contents during international transit. Custom packaging solutions are available upon request to accommodate specific logistical requirements or smaller research quantities.
Every shipment is accompanied by a comprehensive Certificate of Analysis detailing batch-specific test results. Our quality management system ensures full traceability from raw material sourcing to final dispatch. We are committed to providing reliable supply chains for global pharmaceutical partners, ensuring that production schedules are met without compromising on safety or compliance standards. For detailed safety data sheets or technical inquiries, our support team is available to assist with your specific project needs.



