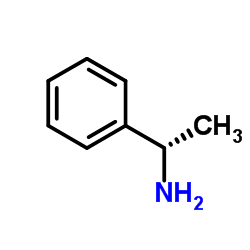
(1R)-1-Phenylethanamine
- CAS No.3886-69-9
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity (1R)-1-phenylethanamine, a chiral amine intermediate widely used in asymmetric synthesis and enantiomeric resolution of acidic compounds.
Request Bulk PricingProduct Technical Details
Product Overview
(1R)-1-Phenylethanamine, also known as (R)-(+)-1-phenylethylamine or R-(+)-α-methylbenzylamine, is a valuable chiral building block in pharmaceutical and fine chemical synthesis. This enantiomerically enriched amine serves as a critical resolving agent for racemic acids and a versatile precursor in the preparation of optically active intermediates. With a molecular formula of C8H11N and a molecular weight of 121.18 g/mol, it is supplied in high optical purity suitable for demanding stereoselective applications.
Specifications
| Appearance | Colorless to light yellow transparent liquid |
|---|---|
| Assay | ≥99.0% (Test result: 99.77%) |
| Specific Rotation | ≥+38.0° (Test result: +39.98°) |
| Optical Purity | ≥98.0% (Test result: 99.08%) |
| Water Content | ≤0.50% (Test result: 0.15%) |
| Molecular Formula | C8H11N |
| Molecular Weight | 121.18 g/mol |
| Density | 0.952 g/mL at 20 °C |
| Boiling Point | 187–189 °C |
| Melting Point | -10 °C |
| Refractive Index | n20/D 1.526 |
| Water Solubility | 40 g/L at 20 °C |
| Storage Conditions | 2–8 °C under inert atmosphere |
Industrial Applications
As a chiral auxiliary and resolving agent, (1R)-1-phenylethanamine plays a pivotal role in the production of enantiopure pharmaceuticals, agrochemicals, and specialty materials. Its primary industrial use involves the diastereomeric salt formation with racemic carboxylic acids to enable separation of enantiomers. Additionally, it serves as a starting material in the synthesis of ligands, catalysts, and complex organic molecules requiring defined stereochemistry. Due to its high enantiomeric excess and stability under standard handling conditions, it is preferred in GMP-compliant manufacturing environments where reproducibility and quality control are paramount.
- Enantiomeric resolution of chiral acids
- Synthesis of asymmetric catalysts and ligands
- Intermediate for active pharmaceutical ingredients (APIs)
- Chiral derivatizing agent in analytical chemistry



