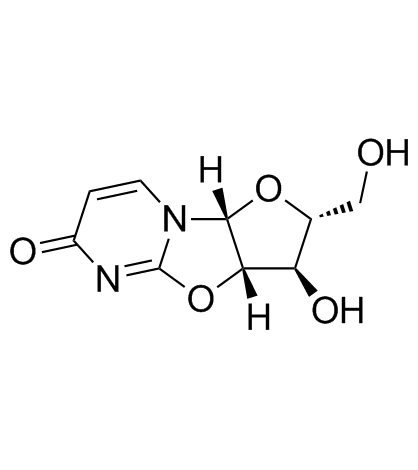
2,2'-O-Anhydro-(1-β-D-Arabinofuranosyl)Uracil
- CAS No.3736-77-4
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity nucleoside intermediate essential for antiviral and anticancer drug synthesis. Available in bulk with full COA.
Request Bulk PricingProduct Technical Details
Product Overview
2,2'-O-Anhydro-(1-β-D-Arabinofuranosyl)Uracil is a specialized nucleoside analog intermediate widely recognized in the pharmaceutical and biochemical research sectors. This compound serves as a critical building block in the synthesis of various nucleoside-based therapeutic agents. Our manufacturing process ensures exceptional chemical stability and purity, making it an ideal choice for complex organic synthesis pathways involving DNA and RNA analogs.
As a key precursor, this material undergoes rigorous quality control measures to meet international standards for pharmaceutical intermediates. The unique anhydro bridge structure provides specific reactivity profiles required for downstream chemical transformations. We supply this product to research institutions and pharmaceutical manufacturers who demand consistency and reliability in their raw materials.
Chemical Specifications
| Parameter | Specification |
|---|---|
| CAS Number | 3736-77-4 |
| Molecular Formula | C9H10N2O5 |
| Molecular Weight | 226.19 g/mol |
| Appearance | White to off-white crystalline powder |
| Assay (HPLC) | ≥99.0% |
| Loss on Drying | ≤0.5% |
| Residue on Ignition | ≤0.1% |
| Heavy Metals | ≤20 ppm |
| Melting Point | 239-240°C |
Industrial Applications
This compound is primarily utilized in the field of medicinal chemistry and nucleoside synthesis. It acts as a vital intermediate for producing cytosine arabinoside derivatives and other antiviral or antineoplastic agents. Researchers leverage its specific stereochemistry to develop new therapeutic candidates targeting viral replication or cellular proliferation.
Beyond direct drug synthesis, this chemical is employed in biochemical assays and drug screening programs. Its high purity ensures minimal interference in sensitive analytical procedures. Laboratories engaged in developing novel nucleoside therapies rely on this intermediate for its consistent performance and reproducible results in multi-step synthesis routes.
Quality Assurance and Storage
We maintain a strict quality management system to guarantee the integrity of every batch. Each shipment is accompanied by a Certificate of Analysis (COA) detailing comprehensive test results. Our production facilities adhere to good manufacturing practices to minimize contamination risks and ensure product safety.
- Strict raw material sourcing and verification
- Advanced HPLC purification techniques
- Comprehensive heavy metal and residue testing
- Stable packaging to prevent moisture absorption
For optimal stability, store this product in a cool, ventilated area away from direct sunlight and moisture. Keep containers tightly sealed when not in use to maintain purity levels. We offer flexible packaging options, including 25 kg drums, to accommodate various scale requirements from pilot studies to full-scale production.



