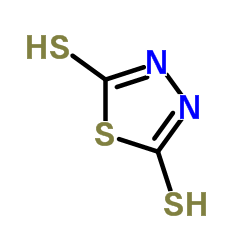
2,5-Dimercapto-1,3,4-thiadiazole
- CAS No.1072-71-5
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity 2,5-Dimercapto-1,3,4-thiadiazole (CAS 1072-71-5), a key pharmaceutical intermediate for cephalosporin antibiotic synthesis such as cefazolin and cefcanel.
Request Bulk PricingProduct Technical Details
Product Overview
2,5-Dimercapto-1,3,4-thiadiazole (CAS 1072-71-5), commonly referenced under the historical name Bismuththiol, is a high-value heterocyclic organic compound widely employed as a critical building block in the pharmaceutical industry. This sulfur-rich molecule serves as an essential intermediate in the synthesis of first-generation cephalosporin antibiotics, including cefazolin and cefcanel. With its dual thiol functionality and stable thiadiazole core, it enables precise molecular construction during complex beta-lactam antibiotic manufacturing.
Specifications
| Molecular Formula | C₂H₂N₂S₃ |
|---|---|
| Molecular Weight | 150.25 g/mol |
| Melting Point | ≥160.0 °C (typical range: 160.3–162.3 °C) |
| Density | 1.8 g/cm³ |
| Boiling Point | 211.3 °C |
| Refractive Index | 1.807 |
| Appearance | Off-white to yellow crystalline powder |
| Assay (Purity) | ≥98.0% (typical result: 98.06%) |
| Loss on Drying | ≤0.5% (typical result: 0.23%) |
| Residue on Ignition | ≤0.5% (typical result: 0.26%) |
Industrial Applications
As a specialized pharmaceutical intermediate, 2,5-Dimercapto-1,3,4-thiadiazole is primarily utilized in the multi-step synthesis of cephalosporin-class antibiotics. Its role is pivotal in constructing the side-chain architecture required for biological activity. Key advantages include:
- High reactivity of dual thiol groups enabling selective derivatization
- Excellent batch-to-batch consistency meeting stringent GMP requirements
- Compatibility with large-scale API manufacturing processes
- Stable under standard storage conditions, ensuring long shelf life
Stored in a cool, well-ventilated area away from oxidizing agents, this compound is typically supplied in 25 kg fiber drums or customized packaging per client specifications. All batches are accompanied by a comprehensive Certificate of Analysis (COA) and full regulatory documentation for seamless integration into global pharmaceutical supply chains.



