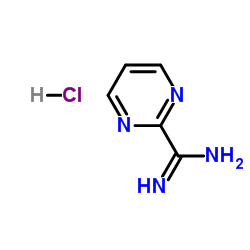
2-Amidinopyrimidine Hydrochloride
- CAS No.138588-40-6
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate used primarily in the synthesis of Bosentan. Available in bulk quantities with strict quality control and comprehensive documentation.
Request Bulk PricingProduct Technical Details
Product Overview
2-Amidinopyrimidine Hydrochloride is a specialized heterocyclic compound that serves as a vital building block in modern pharmaceutical synthesis. Characterized by its stable pyrimidine ring structure combined with an amidine functional group, this chemical exhibits unique reactivity profiles essential for constructing complex therapeutic molecules. As a hydrochloride salt, it offers improved stability and handling properties compared to its free base counterpart, making it a preferred choice for industrial-scale manufacturing processes.
Our facility produces this intermediate under rigorous quality management systems to ensure consistency across batches. The compound is meticulously synthesized to meet high purity standards, minimizing impurities that could affect downstream reactions. This level of quality is critical for clients operating in regulated pharmaceutical environments where trace contaminants must be strictly controlled.
Technical Specifications
| Parameter | Specification |
|---|---|
| Chemical Name | 2-Amidinopyrimidine Hydrochloride |
| CAS Number | 138588-40-6 |
| Molecular Formula | C5H7ClN4 |
| Molecular Weight | 158.589 g/mol |
| Appearance | White to light yellow powder |
| Assay | ≥98.0% |
| Melting Point | 180-183°C (Sublimation) |
| Density | 1.39 g/cm3 |
| Boiling Point | 287.4°C at 760 mmHg |
| Flash Point | 127.6°C |
Industrial Applications
The primary application of 2-Amidinopyrimidine Hydrochloride lies in the synthesis of active pharmaceutical ingredients (APIs), most notably as a key intermediate in the production of Bosentan. Bosentan is an endothelin receptor antagonist used in the treatment of pulmonary arterial hypertension. The precision required in forming the pyrimidine core necessitates high-grade starting materials to ensure the final drug product meets safety and efficacy standards.
Beyond this specific application, the compound serves as a versatile reagent in organic chemistry laboratories. It is utilized in the development of various heterocyclic derivatives where the amidinopyrimidine scaffold provides specific biological activity or structural stability. Researchers value this material for its predictable reaction kinetics and compatibility with various synthetic routes.
Quality Assurance and Handling
We understand that consistency is paramount in chemical manufacturing. Every batch of 2-Amidinopyrimidine Hydrochloride undergoes comprehensive analytical testing, including HPLC, NMR, and melting point analysis, to verify identity and purity. A Certificate of Analysis (COA) is provided with every shipment, detailing the specific test results for that lot number.
- Strict control over raw material sourcing
- In-process quality checks during synthesis
- Final product validation against international standards
- Stability testing to ensure shelf-life integrity
Regarding handling, this compound should be managed in a well-ventilated environment. Personnel are advised to wear appropriate personal protective equipment, including gloves and safety goggles, to prevent irritation. The material is hygroscopic to some extent and should be protected from moisture to maintain optimal physical properties.
Packaging and Storage
To preserve the integrity of the product during transit and storage, we offer robust packaging solutions. The standard package consists of 25 kg drums lined with moisture-resistant bags. However, we recognize that different clients have varying logistical needs. Therefore, we provide customized packaging options upon request to suit specific supply chain requirements.
Storage conditions are critical for maintaining the quality of this hydrochloride salt. The product should be stored in a cool, dry, and ventilated warehouse away from direct sunlight and heat sources. Containers must be kept tightly closed when not in use to prevent exposure to atmospheric moisture. Under recommended storage conditions, the product maintains its specified purity for an extended period, ensuring reliability for long-term production planning.



