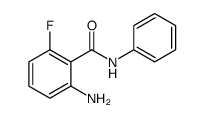
2-Amino-6-fluoro-N-phenylbenzamide
- CAS No.1417456-04-2
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate essential for the synthesis of kinase inhibitors. Manufactured with strict quality control to ensure ≥98% assay reliability for global R&D and production needs.
Request Bulk PricingProduct Technical Details
Product Overview
2-Amino-6-fluoro-N-phenylbenzamide is a specialized organic compound designed for advanced pharmaceutical synthesis. As a critical building block, this fluorinated benzamide derivative plays a pivotal role in the construction of complex therapeutic agents, particularly within the realm of kinase inhibitors. Our manufacturing process emphasizes structural integrity and chemical purity, ensuring that each batch meets the rigorous demands of modern drug discovery and development pipelines.
The incorporation of a fluorine atom at the sixth position of the benzamide ring significantly enhances the metabolic stability and binding affinity of the final active pharmaceutical ingredient. This specific substitution pattern is crucial for optimizing the pharmacokinetic profile of downstream products. We utilize state-of-the-art synthetic routes to achieve consistent quality, making this intermediate a reliable choice for scaling from laboratory research to commercial production.
Technical Specifications
| Parameter | Specification |
|---|---|
| Chemical Name | 2-Amino-6-fluoro-N-phenylbenzamide |
| CAS Number | 1417456-04-2 |
| Molecular Formula | C13H11FN2O |
| Molecular Weight | 230.24 g/mol |
| Appearance | White or off-white powder |
| Assay (Purity) | ≥98.0% |
| Packaging | 25 kg/drum or customized |
Industrial Applications
This compound serves primarily as a key intermediate in the synthesis of Idelalisib, a potent phosphatidylinositol 3-kinase (PI3K) inhibitor used in the treatment of certain hematological malignancies. The high purity level of our product minimizes impurity carry-over during subsequent reaction steps, thereby simplifying purification processes and improving overall yield. Beyond this specific application, the chemical structure offers versatility for medicinal chemists exploring novel analogs in oncology and immunology research.
Our facility supports both standard procurement and custom synthesis projects. We understand that timely delivery and consistent quality are paramount in the pharmaceutical supply chain. Therefore, we maintain robust inventory levels and implement strict batch tracking systems to ensure traceability from raw materials to the final packaged product.
Quality Assurance and Storage
Quality control is integral to our operation. Every production batch undergoes comprehensive analysis using High-Performance Liquid Chromatography (HPLC) and Nuclear Magnetic Resonance (NMR) spectroscopy. A Certificate of Analysis (COA) is provided with each shipment, detailing all relevant physical and chemical properties. We adhere to international safety standards to guarantee that our intermediates are safe for handling in professional laboratory environments.
For optimal stability, the product should be stored in a cool, ventilated area away from direct sunlight and moisture. Containers must be kept tightly sealed when not in use to prevent degradation. We offer flexible packaging options, including 25 kg drums, and can accommodate specific customer requirements for labeling and shipping documentation to facilitate smooth customs clearance and logistics.



