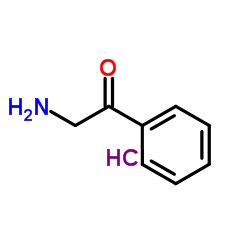
2-Aminoacetophenone Hydrochloride
- CAS No.5468-37-1
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity 2-Aminoacetophenone Hydrochloride (CAS 5468-37-1) designed for pharmaceutical synthesis. Ideal intermediate for Ubenimex production with ≥99% assay.
Request Bulk PricingProduct Technical Details
Product Overview
2-Aminoacetophenone Hydrochloride is a specialized organic compound widely recognized for its critical role in advanced pharmaceutical synthesis. As a stable hydrochloride salt form of 2-aminoacetophenone, this material offers enhanced handling properties and solubility profiles suitable for complex multi-step reaction sequences. Our manufacturing process ensures consistent quality, making it a preferred choice for research laboratories and industrial production facilities focusing on therapeutic agent development.
This chemical building block is particularly valued in the preparation of peptide mimetics and enzyme inhibitors. The structural integrity of the amino ketone framework allows for versatile functionalization, enabling chemists to construct sophisticated molecular architectures required for modern medicinal chemistry programs. We adhere to strict quality control protocols to guarantee that every batch meets the rigorous demands of pharmaceutical-grade applications.
Technical Specifications
| Parameter | Specification |
|---|---|
| Chemical Name | 2-Aminoacetophenone Hydrochloride |
| CAS Number | 5468-37-1 |
| Molecular Formula | C8H10ClNO |
| Molecular Weight | 171.62 g/mol |
| Appearance | Off-white crystalline powder |
| Purity (HPLC) | ≥99.0% |
| Melting Point | 194 °C (dec.) |
| Boiling Point | 247.3 °C at 760 mmHg |
| Density | 1.084 g/cm3 |
Industrial Applications
The primary application of this intermediate lies in the synthesis of Ubenimex, a known aminopeptidase inhibitor used in clinical settings. Beyond this specific pathway, 2-Aminoacetophenone Hydrochloride serves as a versatile precursor in the production of various heterocyclic compounds. Its reactivity profile supports nucleophilic substitutions and condensation reactions, facilitating the creation of diverse chemical libraries for drug discovery initiatives.
Manufacturers utilizing this compound benefit from its high stability under standard storage conditions. The hydrochloride salt form minimizes oxidation risks commonly associated with free base amines, thereby extending shelf life and reducing waste during large-scale processing. This reliability makes it an essential component for supply chains requiring long-term material consistency.
Quality Assurance and Handling
We implement comprehensive quality assurance measures throughout the production lifecycle. Each batch undergoes rigorous analytical testing, including HPLC, NMR, and melting point analysis, to verify identity and purity. A Certificate of Analysis (COA) is provided with every shipment, ensuring full traceability and compliance with international standards.
- Packaged in 25kg cardboard drums or customized containers.
- Stored in a cool, dry, well-closed container away from moisture.
- Protected from strong light and heat sources to maintain stability.
- Recommended storage temperature is refrigerator conditions (+4°C).
Our logistics network supports global delivery, ensuring timely arrival while maintaining product integrity during transit. For specific technical inquiries or custom synthesis requirements, our expert team is available to provide detailed support and documentation.



