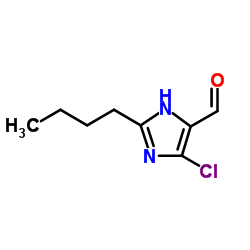
2-Butyl-4-chloro-5-formylimidazole
- CAS No.83857-96-9
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate for Losartan synthesis. Available in bulk with comprehensive quality documentation.
Request Bulk PricingProduct Technical Details
Product Overview
2-Butyl-4-chloro-5-formylimidazole is a critical pharmaceutical intermediate widely recognized for its role in the synthesis of advanced antihypertensive agents. This specialized organic compound features a robust imidazole core substituted with butyl, chloro, and formyl groups, providing the necessary reactivity for downstream coupling reactions. Our manufacturing facility produces this chemical under strict quality control protocols to ensure consistency and reliability for large-scale pharmaceutical production.
As a key building block, this substance serves as a precursor in the construction of complex molecular architectures required for cardiovascular therapeutics. The precise arrangement of functional groups allows for efficient transformation into active pharmaceutical ingredients while maintaining high structural integrity throughout the synthesis pathway.
Technical Specifications
We adhere to rigorous analytical standards to guarantee that every batch meets or exceeds industry expectations for purity and physical properties. The following table outlines the critical parameters verified during our quality assurance process:
| Parameter | Specification |
|---|---|
| Appearance | Off-white to slightly yellow crystalline powder |
| CAS Number | 83857-96-9 |
| Molecular Formula | C8H11ClN2O |
| Molecular Weight | 186.64 g/mol |
| Assay (HPLC) | ≥99.0% |
| Melting Point | 96-99 °C |
| Moisture Content | ≤0.5% |
| Single Impurity | ≤0.15% |
| Total Impurities | ≤1.0% |
Industrial Applications
The primary application of 2-Butyl-4-chloro-5-formylimidazole lies within the pharmaceutical sector, specifically as an intermediate for Losartan synthesis. Losartan is a widely prescribed medication used to manage high blood pressure and protect kidney function in patients with type 2 diabetes. The high purity of our intermediate ensures that the final API meets stringent regulatory requirements for safety and efficacy.
Beyond its primary use, this compound is valuable in research and development settings where imidazole derivatives are required for medicinal chemistry programs. Its stability and reactivity profile make it suitable for various organic synthesis routes involving nucleophilic substitution and condensation reactions.
Quality Assurance and Packaging
Quality is paramount in our production process. Each batch undergoes comprehensive testing using high-performance liquid chromatography (HPLC) and other advanced analytical techniques. We provide a Certificate of Analysis (COA) with every shipment, detailing the test results against our factory standards.
- Consistent batch-to-batch reproducibility
- Low impurity profiles suitable for GMP environments
- Full traceability from raw materials to finished goods
Standard packaging consists of 25 kg drums, designed to protect the product from moisture and light during transit. Custom packaging solutions are available upon request to meet specific logistical requirements. We recommend storing the material in a cool, ventilated area away from direct sunlight to maintain optimal stability over extended periods.
Global Supply Capability
As a established global manufacturer, we possess the capacity to fulfill bulk orders with reliable lead times. Our supply chain is optimized to deliver critical intermediates to pharmaceutical companies and contract manufacturing organizations worldwide. Contact our sales team for current bulk pricing, availability, and technical support regarding synthesis routes.



