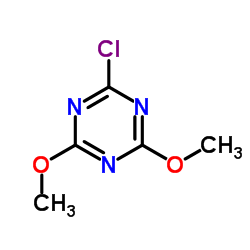
2-Chloro-4,6-dimethoxy-1,3,5-triazine
- CAS No.3140-73-6
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity 2-Chloro-4,6-dimethoxy-1,3,5-triazine (CDMT) serving as an efficient condensation reagent for organic and peptide synthesis.
Request Bulk PricingProduct Technical Details
Product Overview
2-Chloro-4,6-dimethoxy-1,3,5-triazine, commonly known in the industry as CDMT, is a highly specialized heterocyclic compound utilized extensively in advanced organic synthesis. As a premier manufacturer, we supply this critical chemical intermediate with stringent quality controls to ensure optimal performance in complex reaction pathways. This triazine derivative is particularly valued for its role as a condensation reagent, facilitating the formation of amide and ester bonds with high efficiency and minimal side reactions.
Our production facility adheres to international safety and quality standards, delivering a product that meets the rigorous demands of pharmaceutical and fine chemical laboratories. The compound is characterized by its stability and reactivity profile, making it a preferred choice for researchers and industrial chemists seeking reliable coupling agents for multi-step synthesis processes.
Technical Specifications
We maintain strict quality assurance protocols to guarantee the consistency of every batch. The following table outlines the key physical and chemical parameters associated with our standard grade material. Each shipment is accompanied by a comprehensive Certificate of Analysis (COA) verifying these specifications.
| Parameter | Specification |
|---|---|
| Chemical Name | 2-Chloro-4,6-dimethoxy-1,3,5-triazine |
| CAS Number | 3140-73-6 |
| Molecular Formula | C5H6ClN3O2 |
| Molecular Weight | 175.57 g/mol |
| Appearance | White crystal powder |
| Purity (HPLC) | ≥98.0% |
| Melting Point | 71-74°C |
| Density | 1.36 g/cm³ |
| Refractive Index | 1.512 |
Applications and Industrial Benefits
The primary application of this chemical lies in its function as a condensation reagent. It is extensively employed in the activation of carboxylic acids for subsequent nucleophilic attack by amines or alcohols. This mechanism is fundamental in the synthesis of peptides, pharmaceuticals, and specialized polymers. Compared to traditional coupling methods, using this triazine derivative often results in reduced racemization, preserving the stereochemical integrity of sensitive chiral molecules.
- Efficient activation of carboxylic acids under mild conditions.
- Minimizes epimerization during peptide bond formation.
- Compatible with a wide range of organic solvents.
- Suitable for large-scale industrial manufacturing processes.
- High stability during storage ensures long shelf life.
Furthermore, its versatility extends beyond peptide synthesis. It serves as a valuable building block in the development of agrochemicals and functional materials. The high purity level provided by our manufacturing process ensures that downstream reactions proceed with predictable kinetics and high yields, reducing waste and optimizing cost efficiency for our clients.
Storage and Handling Safety
Proper handling and storage are essential to maintain the integrity of this chemical product. The material should be stored in a cool, ventilated area away from direct sunlight and moisture sources. Containers must remain tightly sealed when not in use to prevent hydrolysis or contamination. According to transport regulations, this substance is classified under UN1243 with a hazard class of 3, requiring appropriate packaging and labeling during shipment.
We recommend using personal protective equipment, including gloves and safety goggles, when handling this powder to avoid skin or eye irritation. Our logistics team ensures that all packing requirements, typically 25 kg per drum, are met according to customer specifications and international safety guidelines. For bulk orders or specific packaging needs, our sales team is available to coordinate customized solutions that align with your operational requirements.



