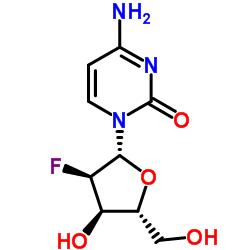
2'-Deoxy-2'-fluorocytidine
- CAS No.10212-20-1
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity 2'-Deoxy-2'-fluorocytidine for antiviral research and pharmaceutical development. Reliable supply with full COA documentation.
Request Bulk PricingProduct Technical Details
Product Overview
2'-Deoxy-2'-fluorocytidine is a specialized fluorinated nucleoside analog that plays a critical role in modern medicinal chemistry and antiviral research. As a key pharmaceutical intermediate, this compound is extensively utilized in the development of novel therapeutic agents targeting viral replication mechanisms. The incorporation of a fluorine atom at the 2'-position of the sugar moiety significantly enhances metabolic stability and biological activity compared to natural nucleosides.
Our manufacturing process ensures exceptional quality and consistency, making this product ideal for laboratory research, preclinical studies, and drug discovery pipelines. We adhere to strict quality control protocols to guarantee that every batch meets rigorous international standards for purity and performance.
Technical Specifications
| Parameter | Specification |
|---|---|
| CAS Number | 10212-20-1 |
| Molecular Formula | C9H12FN3O4 |
| Molecular Weight | 245.21 g/mol |
| Appearance | White to off-white powder |
| Assay (HPLC) | ≥98.0% |
| Loss on Drying | ≤1.0% |
| Heavy Metals | ≤20 ppm |
| Melting Point | 167°C |
Industrial Applications
This fluorinated cytidine derivative is primarily employed as a research tool in virology and oncology. It has demonstrated potent inhibitory activity against specific viral replicons in cell culture models, including hepatitis C virus systems. Researchers utilize this compound to investigate nucleoside metabolism, enzyme kinetics, and structure-activity relationships in the design of next-generation antiviral medications.
- Antiviral drug discovery and development
- In vitro mechanistic studies of viral replication
- Pharmaceutical intermediate for nucleoside analog synthesis
- Quality control reference standard for analytical laboratories
Quality Assurance and Storage
We maintain a robust quality management system to ensure product integrity from synthesis to delivery. Each shipment is accompanied by a comprehensive Certificate of Analysis (COA) detailing batch-specific test results. To maintain stability, the material should be stored in a cool, ventilated environment away from direct sunlight and moisture. Proper handling procedures are recommended to preserve chemical properties during long-term storage.
Our global supply chain capabilities allow us to meet diverse packaging requirements, ranging from standard laboratory quantities to bulk industrial drums. We are committed to supporting our partners with reliable logistics and technical documentation to facilitate seamless research and production workflows.



