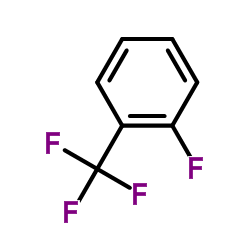
2-Fluorobenzotrifluoride
- CAS No.392-85-8
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity 2-Fluorobenzotrifluoride (CAS 392-85-8) designed for advanced organic synthesis and pharmaceutical intermediate production with guaranteed quality.
Request Bulk PricingProduct Technical Details
Product Overview
2-Fluorobenzotrifluoride is a highly specialized fluorinated aromatic compound that serves as a critical building block in modern organic chemistry. Characterized by the presence of both a fluorine atom and a trifluoromethyl group on the benzene ring, this molecule offers unique reactivity profiles that are essential for the development of advanced pharmaceuticals and agrochemicals. Our manufacturing process ensures exceptional consistency and purity, making it an ideal choice for complex synthetic routes requiring precise substitution patterns.
The ortho-substitution pattern provides specific steric and electronic properties that facilitate nucleophilic aromatic substitution reactions. This makes the compound particularly valuable for researchers and industrial chemists looking to introduce fluorine motifs into larger molecular structures. Fluorination is known to enhance metabolic stability and lipophilicity in drug candidates, thereby increasing the commercial value of downstream products derived from this intermediate.
Key Specifications
| Parameter | Value |
|---|---|
| Chemical Name | 2-Fluorobenzotrifluoride |
| CAS Number | 392-85-8 |
| Molecular Formula | C7H4F4 |
| Molecular Weight | 164.10 g/mol |
| Appearance | Clear colorless Liquid |
| Purity | ≥99.0% |
| Density | 1.293 g/cm3 |
| Boiling Point | 114-115 °C at 750 mm Hg |
| Melting Point | -49 °C |
| Flash Point | 217 °F |
| Refractive Index | 1.406 |
Industrial Applications
This fluorinated intermediate is extensively utilized in the synthesis of trifluoromethoxylated aniline derivatives and other complex organic molecules. Its primary application lies within the pharmaceutical sector, where it acts as a precursor for active pharmaceutical ingredients (APIs) requiring enhanced bioavailability. Additionally, it finds significant use in the agrochemical industry for the production of herbicides and pesticides that demand high environmental stability.
Chemists leverage the reactivity of the fluorine substituent to perform various transformations, including cross-coupling reactions and nucleophilic substitutions. The trifluoromethyl group contributes to the overall electron-withdrawing nature of the molecule, activating the ring for specific chemical modifications. This dual functionality allows for versatile synthetic pathways, reducing the number of steps required to achieve target molecules and improving overall process efficiency.
Quality Assurance and Packaging
We adhere to strict quality control protocols to ensure every batch meets international standards. Each shipment is accompanied by a comprehensive Certificate of Analysis (COA) verifying purity and physical constants. Our facility maintains stable storage conditions to preserve chemical integrity during handling and transit.
- Packaged in 200kg drums or customized containers based on client requirements.
- Stable at room temperature when stored in closed containers.
- Global shipping capabilities with secure handling procedures.
- Technical support available for synthesis route optimization.
Partner with us for reliable supply chain solutions and competitive bulk pricing. Our commitment to quality and safety makes us a preferred global manufacturer for fine chemical intermediates.



