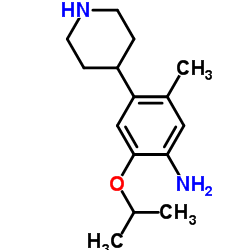
2-Isopropoxy-5-Methyl-4-(Piperidin-4-Yl)Aniline
- CAS No.1035230-24-0
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
Premium grade 2-isopropoxy-5-methyl-4-(piperidin-4-yl)aniline designed for pharmaceutical synthesis. High purity assurance with global supply capabilities.
Request Bulk PricingProduct Technical Details
Product Overview
2-Isopropoxy-5-Methyl-4-(Piperidin-4-Yl)Aniline is a specialized pharmaceutical intermediate characterized by its high chemical stability and precise molecular structure. This compound serves as a critical building block in the synthesis of advanced kinase inhibitors, specifically utilized in the production of Ceritinib. Our manufacturing process ensures consistent quality across batches, making it an ideal choice for research and development teams as well as large-scale commercial production facilities.
As a key component in modern oncology therapeutics, this aniline derivative requires stringent quality control measures. We employ state-of-the-art analytical techniques to verify molecular integrity, ensuring that every shipment meets the rigorous demands of the global pharmaceutical industry. Our commitment to excellence positions us as a reliable partner for complex organic synthesis projects.
Technical Specifications
| Parameter | Value |
|---|---|
| CAS Number | 1035230-24-0 |
| Molecular Formula | C15H24N2O |
| Molecular Weight | 248.364 g/mol |
| Appearance | Yellow powder |
| Assay (HPLC) | ≥99.0% |
| Density | 1.0±0.1 g/cm3 |
| Boiling Point | 390.0±42.0 °C at 760 mmHg |
| Flash Point | 189.7±27.9 °C |
Industrial Applications
The primary application of this chemical lies within the pharmaceutical sector, specifically as an intermediate for Ceritinib synthesis. Ceritinib is a potent inhibitor used in the treatment of non-small cell lung cancer, highlighting the critical nature of this precursor. Beyond oncology, the structural features of this piperidine-aniline compound make it valuable for exploring new therapeutic avenues in medicinal chemistry.
Researchers utilize this intermediate to develop novel formulations requiring high purity standards. The presence of the isopropoxy and methyl groups provides specific steric and electronic properties essential for downstream reactions. Our supply chain supports both pilot-scale experiments and full commercial manufacturing, ensuring continuity for your production timelines.
Quality Assurance and Safety
Quality is paramount in pharmaceutical intermediates. Each batch undergoes comprehensive testing using High-Performance Liquid Chromatography (HPLC) to confirm assay levels exceeding 99.0%. We provide a Certificate of Analysis (COA) with every order, detailing physical and chemical properties to ensure transparency and compliance. Our facilities adhere to strict safety protocols to maintain product integrity during synthesis and packaging.
Handling this compound requires standard laboratory safety precautions. Personnel should use appropriate personal protective equipment, including gloves and eye protection, to prevent exposure. The material should be managed in well-ventilated areas to mitigate any potential risks associated with fine powder handling. We recommend consulting the Safety Data Sheet (SDS) for detailed hazard information and emergency procedures.
Packaging and Logistics
We offer flexible packaging solutions to meet diverse client needs. The standard configuration is 25 kg per drum, secured to prevent contamination during transit. Custom packaging options are available upon request, allowing for tailored quantities that align with your specific inventory requirements. Our logistics network ensures timely global delivery, maintaining optimal storage conditions throughout the shipping process.
Storage recommendations include keeping the product in a cool, ventilated place away from direct sunlight and moisture. Proper storage preserves the chemical stability and extends the shelf life of the material. Our team is available to assist with technical inquiries, providing support from initial sampling to final bulk procurement. Partner with us for reliable supply chain solutions in advanced pharmaceutical intermediates.



