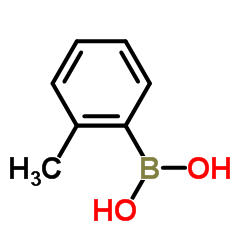
2-Tolylboronic acid
- CAS No.16419-60-6
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity 2-Tolylboronic acid (CAS 16419-60-6) designed for advanced organic synthesis and palladium-catalyzed coupling reactions. Available in bulk quantities with comprehensive COA.
Request Bulk PricingProduct Technical Details
Product Overview
2-Tolylboronic acid, also known systematically as (2-methylphenyl)boronic acid, stands as a critical building block in modern organic synthesis and medicinal chemistry. As a member of the arylboronic acid family, this compound features a boronic acid group attached to the ortho position of a toluene ring. This specific structural arrangement imparts unique reactivity profiles that are highly valued in the construction of complex molecular architectures. Our facility specializes in the production of this key intermediate, ensuring consistent quality and reliability for research and industrial-scale applications.
In the realm of pharmaceutical development and fine chemical manufacturing, the demand for high-purity boronic acids has surged due to their pivotal role in carbon-carbon bond formation. 2-Tolylboronic acid serves as an essential reagent for introducing the o-tolyl moiety into various organic frameworks. Its stability and solubility characteristics make it a preferred choice for chemists working on multi-step synthesis routes where precision and yield are paramount.
Key Specifications
| Parameter | Value |
|---|---|
| Chemical Name | 2-Tolylboronic acid |
| CAS Number | 16419-60-6 |
| Molecular Formula | C7H9BO2 |
| Molecular Weight | 135.96 g/mol |
| Purity | >98% |
| Appearance | White powder |
| Melting Point | 162-164 °C |
| Boiling Point | 283.4 °C at 760 mmHg |
| Density | 1.1 g/cm3 |
Industrial Applications
The primary application of 2-Tolylboronic acid lies in palladium-catalyzed cross-coupling reactions, most notably the Suzuki-Miyaura coupling. This reaction mechanism allows for the efficient synthesis of biaryl compounds, which are ubiquitous structures in active pharmaceutical ingredients (APIs), agrochemicals, and advanced materials. The ortho-methyl substitution can influence the steric environment during coupling, offering chemists specific control over reaction outcomes and selectivity.
Beyond standard coupling reactions, this compound is utilized in the enantiospecific synthesis of diolefins and other complex organic structures. Its utility extends to the development of novel ligands and catalysts where the electronic properties of the boronic acid group are leveraged. Researchers value this intermediate for its ability to facilitate the construction of diverse chemical libraries used in drug discovery programs.
Quality Assurance and Storage
We adhere to strict quality control protocols to ensure that every batch of 2-Tolylboronic acid meets or exceeds industry standards. Each production lot undergoes rigorous testing using advanced analytical techniques such as HPLC and NMR to verify purity and structural integrity. A Certificate of Analysis (COA) is provided with every shipment, detailing the specific test results for your quality assurance records.
- Packaged in 20 kg drums for secure transport.
- Custom packaging solutions available upon request.
- Stored in a sealed container at room temperature.
- Protected from light and moisture to maintain stability.
- Kept in a ventilated and dry environment.
Proper storage is essential to maintain the chemical stability of boronic acids. We recommend keeping the product in its original packaging until ready for use. Our global logistics network ensures timely delivery to laboratories and manufacturing plants worldwide, supporting your production schedules with reliable supply chain management.



