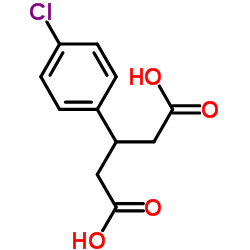
3-(4-Chlorophenyl)pentanedioic Acid
- CAS No.35271-74-0
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity 3-(4-Chlorophenyl)pentanedioic Acid for pharmaceutical synthesis. Reliable bulk supply with comprehensive COA and global shipping capabilities.
Request Bulk PricingProduct Technical Details
Product Overview
3-(4-Chlorophenyl)pentanedioic Acid is a highly specialized organic compound recognized for its critical role in the synthesis of complex pharmaceutical agents. As a key building block, this intermediate facilitates the construction of therapeutic molecules requiring precise stereochemistry and functional group placement. Our manufacturing process ensures exceptional purity levels, making it an ideal choice for research and development teams focused on advanced medicinal chemistry and large-scale production.
The chemical structure features a chlorophenyl moiety attached to a pentanedioic acid backbone, providing unique reactivity profiles essential for multi-step synthesis routes. We prioritize consistency in every batch, adhering to strict international quality standards to support seamless downstream processing. This compound is particularly valued in the preparation of active pharmaceutical ingredients targeting neurological and muscular conditions.
Technical Specifications
Our production facility utilizes advanced analytical techniques to verify every parameter before shipment. The following table outlines the standard physical and chemical properties associated with this high-grade intermediate. Each batch is tested to ensure compliance with the specified assay and physical characteristics.
| Parameter | Value |
|---|---|
| CAS Number | 35271-74-0 |
| Molecular Formula | C11H11ClO4 |
| Molecular Weight | 242.66 g/mol |
| Appearance | White Powder |
| Assay | ≥98.0% |
| Melting Point | 164-166 °C |
| Boiling Point | 394.4 °C |
| Density | 1.4 g/cm3 |
Industrial Applications
This compound serves primarily as a vital precursor in the pharmaceutical industry. It is extensively used in the preparation of active pharmaceutical ingredients APIs targeting specific therapeutic areas. The high purity specification ensures minimal impurity carryover, which is crucial for meeting regulatory compliance in drug manufacturing. Our clients utilize this material for various stages of drug discovery and process development.
- Pharmaceutical intermediate synthesis for therapeutic agents
- Organic chemistry research and development projects
- Custom synthesis projects for specialized chemical structures
- Quality control standard reference in analytical laboratories
Quality Assurance and Storage
We maintain a robust quality management system to guarantee product integrity from synthesis to delivery. Each shipment is accompanied by a Certificate of Analysis COA detailing batch-specific test results including purity, moisture content, and residual solvents. To maintain stability, the product should be stored in a cool, ventilated environment away from direct sunlight and moisture. Proper handling ensures the material retains its specified properties throughout its shelf life.
Personnel handling this chemical should wear appropriate personal protective equipment including gloves and safety goggles. In case of contact with skin or eyes, immediate washing with plenty of water is recommended. Our packaging options include 25 kg drums, with flexibility to accommodate specific customer logistics requirements. Global shipping capabilities ensure timely delivery to laboratories and manufacturing sites worldwide. For technical inquiries or bulk pricing requests, our expert team is available to provide comprehensive support and documentation.



