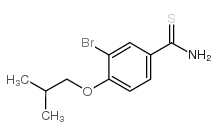
3-Bromo-4-isobutoxybenzothioamide
- CAS No.208665-96-7
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity 3-Bromo-4-isobutoxybenzothioamide (CAS 208665-96-7) designed for pharmaceutical synthesis, specifically as a key intermediate for Febuxostat production.
Request Bulk PricingProduct Technical Details
Product Overview
3-Bromo-4-isobutoxybenzothioamide, identified by CAS number 208665-96-7, represents a critical pharmaceutical intermediate utilized extensively in the synthesis of advanced therapeutic agents. As a specialized organic compound, this substance serves as a foundational building block for the production of Febuxostat, a well-known medication used in the management of hyperuricemia and chronic gout. Our manufacturing facility ensures that every batch meets rigorous international standards, providing chemists and procurement specialists with a reliable source of high-purity materials for research and development purposes.
The chemical structure incorporates a brominated benzothioamide core with an isobutoxy substituent, offering specific reactivity profiles essential for multi-step synthesis routes. Maintaining strict control over impurities is paramount when dealing with intermediates destined for active pharmaceutical ingredient API production. Consequently, our production process emphasizes consistency, stability, and compliance with good manufacturing practices to support downstream pharmaceutical applications effectively.
Technical Specifications
| Parameter | Value |
|---|---|
| Chemical Name | 3-Bromo-4-isobutoxybenzothioamide |
| CAS Number | 208665-96-7 |
| Molecular Formula | C11H14BrNOS |
| Molecular Weight | 288.20 g/mol |
| Appearance | Yellow or yellow-green powder |
| Assay Purity | ≥98.0% |
| Density | 1.398 g/cm3 |
| Boiling Point | 370.783ºC at 760 mmHg |
Industrial Applications
This intermediate is primarily designated for use in the pharmaceutical sector, specifically acting as a key precursor in the manufacturing chain of Febuxostat. The high purity level of ≥98.0% ensures minimal side reactions during subsequent coupling or transformation steps, thereby optimizing overall yield and reducing purification burdens in later stages. Beyond its primary application, this compound may also serve as a valuable reference standard or a building block in the exploration of novel sulfur-containing organic molecules for medicinal chemistry.
- Essential precursor for Febuxostat API synthesis
- Utilized in medicinal chemistry research and development
- Supports scale-up processes for generic pharmaceutical production
- Compatible with various organic synthesis methodologies
Quality Assurance and Storage
Quality control is the cornerstone of our operation. Each production lot undergoes comprehensive analytical testing, including HPLC and NMR verification, to confirm identity and purity levels before release. We provide a Certificate of Analysis COA with every shipment to guarantee transparency and compliance. To maintain product integrity, storage conditions must be strictly observed. The material should be kept in a cool, ventilated environment, away from direct sunlight and moisture sources. Proper packaging in 25 kg drums or customized containers ensures safe transit and long-term stability.
Our commitment extends beyond mere product supply. We offer robust logistical support, ensuring timely delivery to global destinations while adhering to all relevant chemical safety regulations. By choosing our facility, partners gain access to a stable supply chain, competitive bulk pricing, and technical support from experienced chemical engineers dedicated to facilitating your synthesis projects successfully.



