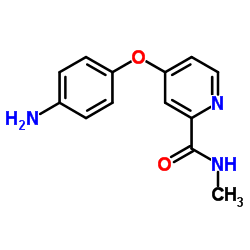
4-(4-Aminophenoxy)-N-methylpyridine-2-carboxamide
- CAS No.284462-37-9
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate for Sorafenib Tosylate synthesis. ≥98% assay, compliant with GMP standards, available for global bulk supply.
Request Bulk PricingProduct Technical Details
Product Overview
4-(4-Aminophenoxy)-N-methylpyridine-2-carboxamide is a specialized organic compound serving as a critical building block in the pharmaceutical industry. Primarily recognized for its role in the synthesis of Sorafenib Tosylate, a multi-kinase inhibitor used in oncology treatments, this intermediate demands stringent quality control and high chemical purity. Our manufacturing process ensures consistent molecular integrity, making it an ideal choice for API production facilities requiring reliable supply chains.
As a key precursor, this compound facilitates the construction of complex therapeutic molecules. The chemical structure combines a pyridine core with an aminophenoxy moiety, offering specific reactivity profiles essential for downstream coupling reactions. We prioritize scalability and reproducibility, supporting both pilot-scale development and commercial manufacturing needs.
Technical Specifications
Our production adheres to rigorous analytical standards to guarantee performance in synthetic routes. The following table outlines the typical quality parameters verified through advanced chromatographic and spectroscopic methods.
| Parameter | Specification |
|---|---|
| CAS Number | 284462-37-9 |
| Molecular Formula | C13H13N3O2 |
| Molecular Weight | 243.26 g/mol |
| Appearance | White to off-white crystalline powder |
| Assay (HPLC) | ≥98.0% |
| Loss on Drying | ≤0.5% |
| Residue on Ignition | ≤0.05% |
| Heavy Metals | ≤10 ppm |
| Single Impurity | ≤0.5% |
| Total Impurities | ≤1.0% |
Quality Assurance and Manufacturing
Quality is embedded in every stage of our production lifecycle. We utilize state-of-the-art synthesis routes optimized for yield and purity. Each batch undergoes comprehensive testing, including Mass Spectrometry (MS), Ultraviolet Spectroscopy (UV), and Proton Nuclear Magnetic Resonance (1H-NMR) for structural confirmation. A Certificate of Analysis (COA) is provided with every shipment, ensuring full traceability and compliance with international regulatory expectations.
Our facility operates under strict environmental and safety protocols. We maintain robust inventory management to ensure timely delivery for global clients. Whether for research purposes or large-scale API manufacturing, our technical team offers support regarding handling, storage, and integration into your specific process workflows.
Industrial Applications
- Primary intermediate for the synthesis of Sorafenib Tosylate (CAS 475207-59-1).
- Utilized in the development of kinase inhibitor pharmaceuticals.
- Applied in medicinal chemistry research for oncology therapeutics.
- Serves as a reference standard for analytical method validation.
Storage and Handling
To maintain stability and potency, this product should be stored in tightly closed containers within a cool, dry, and well-ventilated area. Protection from moisture and direct sunlight is recommended. Standard laboratory safety practices should be observed during handling, including the use of appropriate personal protective equipment. For bulk orders or custom packaging requirements, please contact our sales department for tailored solutions.



