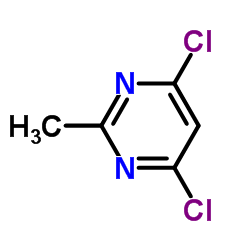
4,6-Dichloro-2-methylpyrimidine
- CAS No.1780-26-3
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity 4,6-Dichloro-2-methylpyrimidine (CAS 1780-26-3) designed for pharmaceutical synthesis. Reliable supply for Dasatinib intermediate production with strict quality control.
Request Bulk PricingProduct Technical Details
Product Overview
4,6-Dichloro-2-methylpyrimidine is a critical heterocyclic building block extensively utilized in the pharmaceutical and agrochemical industries. As a chlorinated pyrimidine derivative, this compound serves as a versatile intermediate for constructing complex molecular architectures. Our manufacturing process ensures high chemical stability and consistent purity, making it an ideal choice for multi-step organic synthesis protocols. We specialize in supplying this material to global partners who require stringent quality standards for active pharmaceutical ingredient (API) production.
Technical Specifications
Quality consistency is paramount in pharmaceutical manufacturing. Our production facilities adhere to rigorous testing protocols to guarantee that every batch meets or exceeds industry standards. The following table outlines the key physical and chemical parameters verified during our quality control process.
| Parameter | Specification |
|---|---|
| CAS Number | 1780-26-3 |
| Molecular Formula | C5H4Cl2N2 |
| Molecular Weight | 163.01 g/mol |
| Appearance | White or off-white powder |
| Purity | ≥98.0% |
| Melting Point | 41.5-45.5 °C |
| Boiling Point | 210.8 °C at 760 mmHg |
| Water Content | ≤0.5% |
| Total Impurities | ≤0.50% |
Industrial Applications
This pyrimidine derivative is primarily recognized for its role in the synthesis of kinase inhibitors. Specifically, it acts as a key intermediate in the production of Dasatinib, a potent therapeutic agent used in oncology treatments. Beyond this specific application, the reactivity of the chloro groups allows for diverse nucleophilic substitution reactions, enabling chemists to introduce various functional groups at the 4 and 6 positions of the pyrimidine ring. This versatility makes it a valuable asset in medicinal chemistry research and development, facilitating the creation of novel drug candidates targeting various biological pathways.
Quality Assurance and Storage
We understand that the integrity of raw materials directly impacts the safety and efficacy of final pharmaceutical products. Therefore, each shipment is accompanied by a comprehensive Certificate of Analysis (COA) detailing batch-specific test results. Our packaging solutions are designed to maintain product stability during transit, typically utilizing 25kg cardboard drums or customized containers based on client requirements. For optimal storage, the material should be kept in a tightly closed container within a cool, dry, and well-ventilated area. It is essential to protect the substance from moisture and incompatible materials to preserve its chemical properties over time. Our logistics team ensures global delivery compliance, adhering to all relevant safety regulations for chemical transport.



