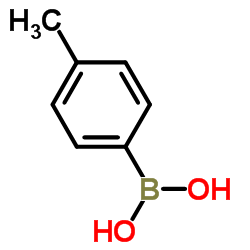
4-Methylphenylboronic Acid
- CAS No.5720-05-8
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity 4-Methylphenylboronic Acid designed for pharmaceutical synthesis and Suzuki-Miyaura cross-coupling reactions.
Request Bulk PricingProduct Technical Details
Product Overview
4-Methylphenylboronic Acid stands as a cornerstone compound in the realm of modern organic synthesis and medicinal chemistry. As a versatile arylboronic acid derivative, this chemical serves as an essential building block for constructing complex molecular architectures. Our facility specializes in the production of this high-grade intermediate, ensuring that every batch meets stringent international quality standards. The compound is widely recognized for its stability and reactivity, making it a preferred choice for research laboratories and industrial manufacturing plants alike.
In the context of pharmaceutical development, the reliability of raw materials is paramount. We understand that the integrity of the final active pharmaceutical ingredient (API) depends heavily on the purity of the starting materials. Therefore, our manufacturing process is designed to minimize impurities and maximize yield consistency. This commitment to quality allows our partners to streamline their synthesis workflows without compromising on safety or efficacy.
Key Specifications
| Parameter | Specification |
|---|---|
| Product Name | 4-Methylphenylboronic Acid |
| CAS Number | 5720-05-8 |
| Molecular Formula | C7H9BO2 |
| Molecular Weight | 135.96 g/mol |
| Appearance | White powder |
| Assay | ≥99.0% |
| Water Content | ≤0.50% |
| Melting Point | 256-263 °C |
| UN Number | UN2206 |
Industrial Applications
The primary utility of 4-Methylphenylboronic Acid lies in its capability to facilitate carbon-carbon bond formation through palladium-catalyzed cross-coupling reactions. Specifically, it is extensively utilized in Suzuki-Miyaura coupling processes involving aryl bromides. This reaction mechanism is fundamental in the synthesis of biaryl structures, which are prevalent in numerous therapeutic agents. Beyond standard coupling, this reagent is also effective in palladium-catalyzed direct arylation, offering chemists a robust tool for diversifying molecular scaffolds.
Our clients leverage this intermediate for various high-value applications, including the development of novel pharmaceuticals, agrochemicals, and advanced functional materials. The high purity level ensures that side reactions are minimized, leading to cleaner reaction profiles and easier downstream purification. Whether used in small-scale discovery chemistry or large-scale commercial production, this boronic acid derivative delivers consistent performance.
Quality Assurance and Benefits
- High Purity Grade: Assay guaranteed at 99.0% or higher for optimal reaction efficiency.
- Rigorous Testing: Every batch undergoes strict quality control including HPLC and moisture analysis.
- Global Supply Chain: Reliable logistics network ensuring timely delivery to international destinations.
- Technical Support: Dedicated team available to assist with synthesis route optimization.
- Custom Packaging: Flexible options available to meet specific production line requirements.
Storage and Handling
Proper storage is critical to maintaining the chemical stability of 4-Methylphenylboronic Acid. The product should be stored in a cool, dry, and well-ventilated area away from direct sunlight and moisture sources. Containers must remain tightly closed when not in use to prevent hydration or contamination. As classified under UN2206 with a transport hazard class of 6.1, appropriate safety measures must be followed during shipping and handling. We provide comprehensive safety data sheets (SDS) and certificates of analysis (COA) with every shipment to ensure full regulatory compliance and workplace safety.



