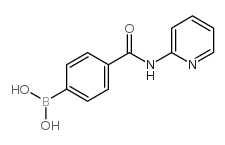
4-(Pyridin-2-yl)aminocarbonylphenylboronic acid
- CAS No.850568-25-1
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity 4-(Pyridin-2-yl)aminocarbonylphenylboronic acid designed for kinase inhibitor synthesis. Reliable supply with full COA documentation.
Request Bulk PricingProduct Technical Details
Product Overview
4-(Pyridin-2-yl)aminocarbonylphenylboronic acid is a specialized organic building block extensively utilized in the pharmaceutical industry for the synthesis of complex kinase inhibitors. As a critical intermediate, this compound plays a pivotal role in constructing the molecular framework required for advanced therapeutic agents. Our manufacturing process ensures exceptional consistency and purity, meeting the rigorous demands of modern drug discovery and development pipelines.
This boronic acid derivative is particularly valued for its stability and reactivity in cross-coupling reactions, such as Suzuki-Miyaura couplings, which are fundamental in assembling biaryl structures found in many oncology treatments. We maintain strict quality control protocols to guarantee that every batch supports efficient downstream synthesis without compromising yield or purity.
Technical Specifications
| Parameter | Value |
|---|---|
| Chemical Name | 4-(Pyridin-2-yl)aminocarbonylphenylboronic acid |
| CAS Number | 850568-25-1 |
| Molecular Formula | C12H11BN2O3 |
| Molecular Weight | 242.04 g/mol |
| Purity | ≥98.0% |
| Appearance | White to off-white solid |
| Melting Point | 124-128°C |
| Density | 1.33 g/cm³ |
Industrial Applications
The primary application of this compound lies in the synthesis of pharmaceutical intermediates, specifically serving as a key precursor for kinase inhibitors like Acalabrutinib. Its structural properties enable precise functionalization, making it indispensable for medicinal chemists aiming to develop targeted cancer therapies. Beyond oncology, boronic acid derivatives are increasingly explored in various therapeutic areas due to their unique binding capabilities.
Our clients utilize this material for process development, scale-up, and commercial manufacturing. The high purity level minimizes impurity profiles in the final active pharmaceutical ingredient (API), ensuring regulatory compliance and patient safety. We support R&D teams with reliable supply chains that accommodate both small-scale laboratory needs and large-scale production requirements.
Quality Assurance and Packaging
Quality is paramount in pharmaceutical chemical manufacturing. Each production lot undergoes comprehensive analysis using HPLC, NMR, and mass spectrometry to verify identity and purity. A Certificate of Analysis (COA) is provided with every shipment, detailing all critical quality attributes. Our facility adheres to international safety and environmental standards, ensuring responsible production practices.
- Strict quality control testing for every batch
- Comprehensive documentation including COA and MSDS
- Stable supply chain for global distribution
- Custom packaging solutions available upon request
Standard packaging includes 25 kg drums, securely sealed to prevent moisture absorption and degradation. Storage recommendations advise keeping the product in a cool, ventilated area away from direct sunlight and incompatible materials. For specific packaging needs or technical inquiries regarding synthesis routes, our expert team is available to provide detailed support and guidance.



