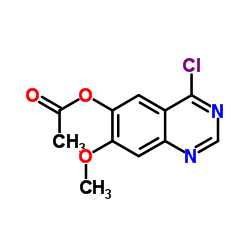
6-Acetoxy-4-Chloro-7-Methoxyquinazoline
- CAS No.230955-75-6
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity 6-Acetoxy-4-Chloro-7-Methoxyquinazoline (CAS 230955-75-6) available for pharmaceutical synthesis. Reliable global supplier offering competitive bulk pricing and full documentation.
Request Bulk PricingProduct Technical Details
Product Overview
6-Acetoxy-4-Chloro-7-Methoxyquinazoline is a specialized quinazoline derivative primarily utilized in the pharmaceutical industry as a critical building block. This compound serves as a key intermediate in the multi-step synthesis of advanced kinase inhibitors, specifically contributing to the production of targeted oncology therapeutics. Our manufacturing facility ensures that every batch meets stringent international standards for chemical purity and structural integrity, supporting reliable downstream reactions for research and development teams.
The molecular structure of this quinazoline derivative features specific substitution patterns that enhance its reactivity in nucleophilic substitution processes. These chemical characteristics make it an indispensable precursor for constructing complex heterocyclic frameworks required in modern drug discovery pipelines. By maintaining precise control over the acetoxy and chloro functional groups, we guarantee consistent performance during the synthesis of active pharmaceutical ingredients.
Technical Specifications
Quality control is paramount in the production of pharmaceutical intermediates. Our comprehensive testing protocols verify every parameter to ensure compliance with global regulatory requirements. The following table outlines the standard specifications for this product:
| Parameter | Specification |
|---|---|
| CAS Number | 230955-75-6 |
| Molecular Formula | C11H9ClN2O3 |
| Molecular Weight | 252.65 g/mol |
| Appearance | Off-white or white powder |
| Purity | ≥98.0% |
| Individual Impurity | ≤0.5% |
| Total Impurities | ≤1.0% |
| Loss on Drying | ≤0.5% |
| Residue on Ignition | ≤0.1% |
| Heavy Metals | ≤10 ppm |
Industrial Applications
This intermediate is predominantly employed in the manufacturing process of Gefitinib and related tyrosine kinase inhibitors. These medications are vital in the treatment of various forms of cancer, highlighting the importance of high-quality raw materials. Pharmaceutical companies rely on consistent supply chains to maintain production schedules for clinical trials and commercial manufacturing. Our product facilitates efficient synthesis routes, reducing waste and improving overall yield in the final API production.
Quality Assurance and Safety
We adhere to rigorous quality management systems to ensure product safety and reliability. Each shipment is accompanied by a Certificate of Analysis (COA) detailing batch-specific test results. Our logistics team ensures proper packaging to prevent degradation during transit. For safety, this chemical should be handled in a well-ventilated area with appropriate personal protective equipment. Storage conditions require a cool, dry environment away from direct light and moisture to maintain stability over extended periods.
Key Advantages
- Consistent high purity levels suitable for GMP manufacturing
- Competitive bulk pricing for large-scale production needs
- Global shipping capabilities with secure packaging
- Full technical support and documentation provided
- Strict compliance with environmental and safety regulations



