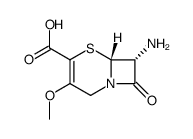
7-Amino-3-methoxy-3-cephem-4-carboxylic acid
- CAS No.51803-38-4
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity 7-Amino-3-methoxy-3-cephem-4-carboxylic acid intermediate for cephalosporin antibiotic synthesis, available in bulk with full COA.
Request Bulk PricingProduct Technical Details
Product Overview
7-Amino-3-methoxy-3-cephem-4-carboxylic acid, commonly known as 7-AMOCA, is a critical pharmaceutical intermediate utilized extensively in the synthesis of cephalosporin antibiotics. This compound serves as a foundational building block for producing advanced therapeutic agents, specifically playing a pivotal role in the manufacturing of Cefroxadine. Our facility ensures that every batch meets stringent international quality standards, providing reliable supply chains for global pharmaceutical manufacturers.
As a key cephem nucleus derivative, this chemical exhibits exceptional stability and reactivity profiles required for complex organic synthesis. The presence of the methoxy group at the 3-position enhances the beta-lactamase stability of the final antibiotic products, making this intermediate highly valuable in modern medicinal chemistry. We specialize in the large-scale production of this compound, adhering to Good Manufacturing Practices (GMP) to guarantee consistency and safety.
Chemical Properties and Structure
The molecular structure of 7-Amino-3-methoxy-3-cephem-4-carboxylic acid features a bicyclic beta-lactam system fused with a dihydrothiazine ring. This specific configuration, designated as (6R,7R)-7-amino-3-methoxy-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid, is essential for maintaining the biological activity of downstream antibiotics. The compound is characterized by its high molecular integrity and low impurity profile, which are crucial for minimizing side reactions during subsequent coupling steps.
Physically, the substance presents as a white to yellow powder, indicating high purity levels when processed correctly. Its thermal stability and solubility characteristics are optimized for industrial-scale reactions, allowing for efficient processing in various solvent systems. Our quality control teams perform rigorous testing on each batch to verify molecular weight, density, and functional group integrity.
Industrial Applications
The primary application of this intermediate is in the semi-synthetic production of cephalosporin antibiotics. It acts as a core scaffold upon which various side chains are attached to create specific drug molecules. Beyond Cefroxadine, this building block is increasingly relevant in the development of next-generation antibiotics designed to combat resistant bacterial strains. Pharmaceutical companies rely on this intermediate for its consistent performance in acylation and protection-deprotection sequences.
- Essential precursor for Cefroxadine synthesis
- Used in developing broad-spectrum cephalosporins
- Supports research into beta-lactamase resistant antibiotics
- Compatible with standard industrial coupling reagents
Specifications and Quality Assurance
We maintain a robust quality assurance framework to ensure that all shipped products meet or exceed the specified purity requirements. Our analytical laboratories utilize high-performance liquid chromatography (HPLC) and nuclear magnetic resonance (NMR) to validate the chemical identity and purity of every lot. The following table outlines the standard technical specifications for our commercial grade material.
| Parameter | Specification |
|---|---|
| CAS Number | 51803-38-4 |
| Molecular Formula | C8H10N2O4S |
| Molecular Weight | 230.24 g/mol |
| Appearance | White to yellow powder |
| Purity | ≥99.0% |
| Density | 1.64 g/cm3 |
| Boiling Point | 552.8 °C |
| Flash Point | 288.1 °C |
Packaging and Storage
To preserve the chemical stability of 7-Amino-3-methoxy-3-cephem-4-carboxylic acid, proper packaging and storage conditions are strictly enforced. The product is typically supplied in 25 kg drums, lined with moisture-proof bags to prevent degradation from humidity. Custom packaging solutions are available upon request to accommodate specific logistical requirements or smaller scale research needs.
Storage should be maintained in a cool, ventilated area away from direct sunlight and strong oxidizing agents. Under recommended conditions, the product retains its specified purity for extended periods. We provide comprehensive documentation with every shipment, including Certificates of Analysis (COA), Material Safety Data Sheets (MSDS), and method validation reports to support regulatory filings.



