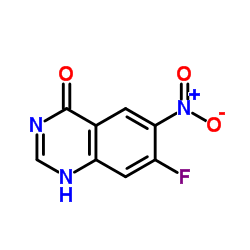
7-Fluoro-6-Nitro-4-Hydroxyquinazoline
- CAS No.162012-69-3
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity 7-Fluoro-6-Nitro-4-Hydroxyquinazoline (CAS 162012-69-3) serves as a critical pharmaceutical intermediate for Afatinib synthesis. Available in bulk quantities with strict quality control.
Request Bulk PricingProduct Technical Details
Product Overview
7-Fluoro-6-Nitro-4-Hydroxyquinazoline is a specialized heterocyclic compound widely recognized for its pivotal role in modern pharmaceutical synthesis. As a key quinazoline derivative, this chemical building block is essential for the development of advanced kinase inhibitors, particularly in the production of oncology therapeutics such as Afatinib. Our manufacturing process ensures consistent molecular integrity and high purity levels, meeting the rigorous demands of global pharmaceutical research and production facilities.
We understand that the quality of intermediates directly impacts the efficacy and safety of the final drug product. Therefore, our facility adheres to strict quality management systems to deliver material that supports seamless downstream synthesis. Whether for process development or large-scale commercial manufacturing, this compound provides a reliable foundation for complex organic synthesis routes.
Key Specifications
| Parameter | Specification |
|---|---|
| Chemical Name | 7-Fluoro-6-Nitro-4-Hydroxyquinazoline |
| CAS Number | 162012-69-3 |
| Molecular Formula | C8H4FN3O3 |
| Molecular Weight | 209.13 g/mol |
| Purity (HPLC) | ≥98.0% |
| Appearance | White to yellow to light brown solid |
| Boiling Point | 136°C |
| Flash Point | 37°C |
Industrial Applications
This compound is primarily utilized as a critical intermediate in the pharmaceutical industry. Its specific structural configuration allows for efficient coupling reactions required in the synthesis of epidermal growth factor receptor (EGFR) inhibitors. Researchers and process chemists value this material for its reactivity profile and stability under standard storage conditions.
- Essential building block for Afatinib and related kinase inhibitors.
- Used in medicinal chemistry for structure-activity relationship (SAR) studies.
- Supports large-scale commercial production of targeted cancer therapies.
- Compatible with various nucleophilic substitution reactions.
Quality Assurance and Storage
Quality control is paramount in our supply chain. Each batch undergoes comprehensive testing using high-performance liquid chromatography (HPLC) to verify purity and identity. A Certificate of Analysis (COA) is provided with every shipment to ensure transparency and compliance with your internal quality standards. We maintain full traceability from raw material sourcing to final packaging.
To maintain optimal stability, the product should be stored in a cool, ventilated area away from direct sunlight and moisture. Proper storage conditions prevent degradation and ensure the material remains within specification until use. Our packaging solutions are designed to protect the integrity of the chemical during transit and long-term storage.
Packaging and Logistics
We offer flexible packaging options to suit diverse operational needs. The standard packaging configuration is 25 kg per drum, which balances ease of handling with economic shipping efficiency. For clients with specific requirements, custom packaging solutions are available upon request. Our logistics team ensures timely global delivery while adhering to all relevant transportation regulations, including proper hazard classification (UN1987, Hazard Class 3).
Partner with us for a reliable supply of high-quality pharmaceutical intermediates. Our commitment to technical excellence and customer support makes us a preferred choice for manufacturers worldwide.



