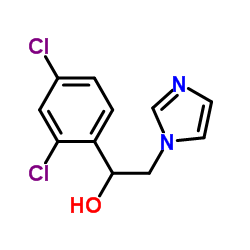
α-(2,4-Dichlorophenyl)-1H-imidazole-1-ethanol
- CAS No.24155-42-8
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate essential for the synthesis of antifungal agents like Miconazole Nitrate, available in bulk quantities with full quality documentation.
Request Bulk PricingProduct Technical Details
Product Overview
α-(2,4-Dichlorophenyl)-1H-imidazole-1-ethanol is a specialized organic compound serving as a critical building block in the pharmaceutical industry. This imidazole derivative is primarily utilized in the manufacturing of broad-spectrum antifungal medications. Our production facility adheres to strict international quality standards, ensuring that every batch meets the rigorous demands of modern drug synthesis. The compound is characterized by its stable chemical structure and high reactivity, making it an ideal precursor for complex azole-based therapeutic agents.
As a leading global manufacturer, we understand the importance of consistency in pharmaceutical supply chains. Our synthesis process is optimized to minimize impurities and maximize yield, providing our partners with a reliable source of high-grade intermediates. Whether for research and development or large-scale commercial production, this material is processed under controlled conditions to maintain integrity and performance.
Technical Specifications
| Parameter | Specification |
|---|---|
| CAS Number | 24155-42-8 |
| Molecular Formula | C11H10Cl2N2O |
| Molecular Weight | 257.116 g/mol |
| Appearance | Light brown to white crystalline powder |
| Assay (HPLC) | ≥99.0% |
| Assay (Titration) | 98.5-101.5% |
| Melting Point | 134-138 °C |
| Single Impurity | ≤0.14% |
| Total Impurities | ≤1.0% |
| Loss on Drying | ≤0.2% |
| Sulfated Ash | ≤0.1% |
| Density | 1.4±0.1 g/cm3 |
| Boiling Point | 468.5±45.0 °C at 760 mmHg |
| Flash Point | 237.1±28.7 °C |
Industrial Applications
The primary application of this chemical is in the synthesis of Miconazole Nitrate, a well-known antifungal agent used to treat various fungal infections. The high purity of our intermediate ensures that the downstream reaction processes proceed efficiently, reducing waste and improving the overall quality of the final active pharmaceutical ingredient (API). Beyond antifungal medications, imidazole derivatives are increasingly explored in other therapeutic areas, including potential applications in agrochemicals and specialized material science.
Our clients benefit from a versatile product that integrates seamlessly into existing synthesis routes. The chemical properties, including its specific melting point and solubility profile, are tightly controlled to facilitate predictable reaction kinetics. This reliability is crucial for pharmaceutical manufacturers who must adhere to strict regulatory guidelines during drug production.
Quality Assurance and Storage
Quality is the cornerstone of our manufacturing philosophy. Each production batch undergoes comprehensive testing using advanced analytical techniques such as High-Performance Liquid Chromatography (HPLC) and titration. We provide a Certificate of Analysis (COA) with every shipment, detailing all critical quality attributes. Our facility operates under Good Manufacturing Practices (GMP) to ensure safety and consistency.
- Packaging: Standard 25 kg drums, with custom packaging available upon request.
- Storage: Store in a cool, ventilated place away from direct sunlight and moisture.
- Stability: Stable under recommended storage conditions for extended periods.
- Support: Dedicated technical support team available for formulation guidance.
We are committed to supplying the global market with safe, compliant, and high-performance chemical intermediates. Partner with us for reliable supply chains and competitive bulk pricing structures tailored to your production needs.



