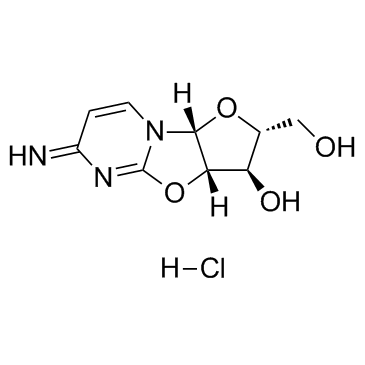
Ancitabine Hydrochloride
- CAS No.10212-25-6
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Ancitabine Hydrochloride (CAS 10212-25-6) designed for pharmaceutical synthesis and ophthalmic formulations.
Request Bulk PricingProduct Technical Details
Product Overview
Ancitabine Hydrochloride is a specialized nucleoside analog derivative widely recognized within the pharmaceutical industry for its utility in complex organic synthesis. As a critical chemical building block, this compound serves as a foundational intermediate in the development of advanced therapeutic agents. Our facility produces this material under stringent quality control protocols to ensure consistency across batches, catering to the rigorous demands of pharmaceutical research and manufacturing sectors.
The chemical structure features a modified cytidine backbone, which provides unique reactivity profiles essential for downstream synthesis processes. Professionals in medicinal chemistry value this compound for its stability and defined physical properties, making it a reliable choice for scaling up production of specialized medications.
Technical Specifications
We maintain high standards for purity and physical characteristics to support reliable experimental outcomes and manufacturing efficiency. The following table outlines the key technical parameters verified through our quality assurance laboratory.
| Parameter | Specification |
|---|---|
| CAS Number | 10212-25-6 |
| Molecular Formula | C9H12ClN3O4 |
| Molecular Weight | 261.66 g/mol |
| Appearance | White or off-white crystalline powder |
| Assay (HPLC) | ≥98.0% |
| Melting Point | 269-270°C |
| Density | 2.01 g/cm³ |
Quality Assurance and Testing
Every batch of Ancitabine Hydrochloride undergoes comprehensive analytical testing before release. We utilize high-performance liquid chromatography (HPLC) to verify assay levels, ensuring that impurity profiles remain within acceptable limits for pharmaceutical intermediate use. Our quality management system documents all testing results, and a Certificate of Analysis (COA) is provided with every shipment to guarantee traceability and compliance.
Storage stability is monitored to prevent degradation. The compound should be kept in a cool, ventilated environment away from direct sunlight and moisture to maintain its chemical integrity over time. Proper handling procedures are recommended to ensure safety during laboratory or industrial processing.
Industrial Applications
This chemical is primarily utilized as a key intermediate in the synthesis of anti-tumor pharmaceutical agents. Its structural properties make it suitable for incorporation into complex molecular frameworks required for oncology research. Additionally, it finds application in the formulation of ophthalmic solutions, where high purity is paramount to ensure safety and efficacy in final products.
Our supply chain supports both research-scale quantities and bulk industrial requirements. We work closely with clients to understand their specific synthesis routes, offering technical support to optimize the integration of this intermediate into their manufacturing processes.
Packaging and Logistics
Standard packaging consists of 25 kg drums, designed to protect the material from environmental factors during transit. Custom packaging solutions are available upon request to meet specific logistical needs. We coordinate global shipping with careful attention to regulatory requirements, ensuring timely delivery to laboratories and production facilities worldwide.



