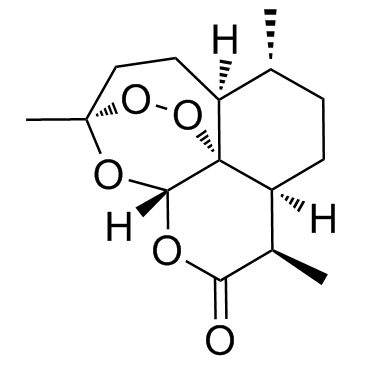
Artemisinin
- CAS No.63968-64-9
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity Artemisinin API intermediate for antimalarial formulations with strict quality control and global supply capabilities.
Request Bulk PricingProduct Technical Details
Product Overview
Artemisinin represents a critical advancement in modern pharmaceutical chemistry, recognized globally for its efficacy in therapeutic applications. This sesquiterpene lactone contains a unique peroxide bridge structure that is essential for its biological activity. Our facility specializes in the production of high-grade Artemisinin, ensuring consistent quality for pharmaceutical manufacturers and research institutions worldwide. We adhere to stringent manufacturing protocols to deliver a product that meets international standards for purity and safety.
The compound is derived through advanced extraction and purification processes from natural sources, followed by rigorous refinement to achieve pharmaceutical grade specifications. Our commitment to excellence ensures that every batch maintains the structural integrity required for downstream synthesis and formulation. As a trusted supplier, we understand the vital role this chemical plays in global health initiatives and dedicate our resources to maintaining a reliable supply chain.
Technical Specifications
Quality is paramount in the production of fine chemicals and active pharmaceutical ingredients. The following table outlines the key physical and chemical properties of our Artemisinin product, verified through comprehensive analytical testing.
| Parameter | Specification |
|---|---|
| CAS Number | 63968-64-9 |
| Molecular Formula | C15H22O5 |
| Molecular Weight | 282.33 g/mol |
| Appearance | White to off-white crystalline powder |
| Assay (HPLC) | 98.0% - 102.0% |
| Melting Point | 156-157°C |
| Loss on Drying | ≤0.5% |
| Residue on Ignition | ≤0.1% |
| Total Impurities | ≤1.0% |
Quality Control and Assurance
Our quality assurance framework employs state-of-the-art analytical techniques to verify product identity and purity. Each production lot undergoes extensive testing using High-Performance Liquid Chromatography (HPLC) to quantify the main component and detect related substances. We monitor specific impurities such as Artemisitene and epimers to ensure they remain within acceptable limits defined by pharmacopoeia standards.
Heavy metal analysis is conducted regularly to confirm compliance with safety regulations regarding Lead, Arsenic, Mercury, and Cadmium. Microbiological testing ensures the product is free from harmful contaminants including Escherichia coli, Salmonella, and Staphylococcus aureus. A Certificate of Analysis (COA) is provided with every shipment, documenting all test results against our factory standards.
Industrial Applications
Artemisinin serves as a foundational building block in the pharmaceutical industry, primarily utilized in the development of antimalarial medications. It acts as a key intermediate for various derivatives used in combination therapies. Beyond direct pharmaceutical use, it is also employed in research settings for studying organic synthesis pathways involving peroxide structures.
Our clients include established pharmaceutical companies and biotechnology firms requiring reliable raw materials for formulation. The high purity profile of our product makes it suitable for sensitive manufacturing processes where consistency is critical. We support our partners with technical data and regulatory documentation to facilitate smooth integration into their production lines.
Storage and Stability
Proper storage conditions are essential to maintain the stability and efficacy of Artemisinin. The product should be kept in a ventilated, low-temperature, and dry environment to prevent degradation. It is stable under normal conditions but is combustible and should be kept away from strong oxidizing agents, acids, acid chlorides, and acid anhydrides.
Packaging is designed to protect the material from moisture and light exposure during transit. We recommend storing the containers in a secure warehouse facility with controlled humidity. Handling procedures should follow standard safety protocols for fine chemicals, including the use of appropriate personal protective equipment to ensure operator safety during weighing and processing operations.



