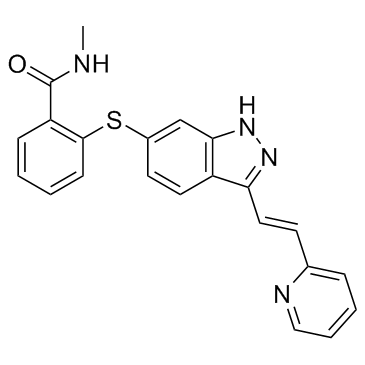
Axitinib
- CAS No.319460-85-0
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
Axitinib is a high-purity tyrosine kinase inhibitor intermediate designed for pharmaceutical research and large-scale API manufacturing processes.
Request Bulk PricingProduct Technical Details
Product Overview
Axitinib represents a critical advancement in the field of oncology pharmaceuticals, serving as a potent multi-target tyrosine kinase inhibitor. Our facility specializes in the production of high-purity Axitinib intermediates, catering to the rigorous demands of global pharmaceutical manufacturers and research institutions. This compound is meticulously synthesized to ensure optimal bioavailability and efficacy when formulated into final dosage forms. As a key component in targeted cancer therapies, maintaining exceptional chemical integrity is paramount for downstream applications.
The chemical structure of Axitinib, characterized by its indazole and pyridine moieties, requires precise synthetic control to maintain the correct stereochemistry, specifically the (1E)-ethenyl configuration. Our manufacturing processes are designed to uphold these structural necessities, ensuring that every batch meets the stringent requirements necessary for clinical and commercial drug production. We understand the critical nature of supply chain reliability in the pharmaceutical sector and commit to consistent quality across all shipments.
Chemical Properties and Specifications
Our Axitinib intermediate is produced under strict quality control protocols, aligning with international pharmacopoeia standards. The physical properties are carefully monitored to ensure compatibility with various formulation techniques. The compound exhibits specific solubility characteristics that must be accounted for during the drug development phase. Below is a detailed summary of the key specification parameters verified through our analytical laboratories.
| Parameter | Specification | Typical Result |
|---|---|---|
| Appearance | Pale-yellow to off-white powder | Off-white powder |
| CAS Number | 319460-85-0 | 319460-85-0 |
| Molecular Formula | C22H18N4OS | C22H18N4OS |
| Molecular Weight | 386.47 g/mol | 386.47 g/mol |
| Assay (HPLC) | 98.0% - 102.0% | 99.96% |
| Total Impurities | ≤1.0% | 0.33% |
| Loss on Drying | ≤1.0% | 0.45% |
| Residue on Ignition | ≤0.1% | 0.06% |
Pharmacological Mechanism
From a biochemical perspective, Axitinib functions by selectively inhibiting vascular endothelial growth factor receptors (VEGFR1, VEGFR2, and VEGFR3) as well as platelet-derived growth factor receptor beta (PDGFRβ). This inhibition mechanism disrupts the signaling pathways responsible for angiogenesis, effectively starving tumor cells of the blood supply required for growth and metastasis. The IC50 values for these receptors are in the low nanomolar range, demonstrating high potency. For pharmaceutical developers, understanding this mechanism is crucial when designing combination therapies or assessing potential drug interactions.
Quality Assurance and Manufacturing
Quality assurance is the cornerstone of our production philosophy. Every batch of Axitinib undergoes comprehensive testing using validated methods including High-Performance Liquid Chromatography (HPLC), Ultraviolet Spectroscopy (UV), and mass spectrometry. We maintain full traceability from raw material sourcing to final packaging. Our facilities adhere to Good Manufacturing Practices (GMP) guidelines, ensuring that the intermediates supplied are suitable for use in regulated pharmaceutical environments. Certificates of Analysis (COA) are provided with every shipment to verify compliance with agreed specifications.
Industrial Applications
The primary application of our Axitinib intermediate is in the manufacturing of finished pharmaceutical products for oncology treatment. It serves as the Active Pharmaceutical Ingredient (API) for tablets and capsules intended for clinical use. Additionally, it is utilized in preclinical research to study kinase inhibition pathways and develop novel therapeutic combinations. Our clients range from large-scale pharmaceutical corporations to specialized biotechnology firms engaged in innovative cancer research.
- Large-scale API manufacturing for oral solid dosage forms.
- Preclinical research and development in oncology.
- Reference standard preparation for analytical laboratories.
- Formulation studies for improved bioavailability.
Safety and Handling
While Axitinib is a potent pharmaceutical compound, it must be handled with appropriate safety measures during industrial processing. Personnel should utilize standard personal protective equipment (PPE) including gloves, safety goggles, and respiratory protection when handling powders to prevent inhalation or skin contact. Storage conditions should maintain a cool, dry environment away from direct sunlight to preserve chemical stability. Proper waste disposal protocols must be followed in accordance with local environmental regulations.



