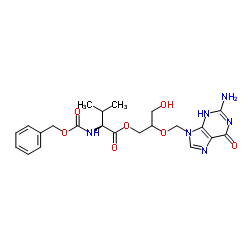
CBZ-Valganciclovir
- CAS No.194154-40-0
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
CBZ-Valganciclovir is a high-purity pharmaceutical intermediate essential for antiviral drug synthesis. We provide reliable supply with strict quality control and comprehensive documentation.
Request Bulk PricingProduct Technical Details
Product Overview
CBZ-Valganciclovir, identified by CAS number 194154-40-0, serves as a critical protected intermediate in the pharmaceutical industry. This compound functions as a key building block in the synthesis of advanced antiviral agents. Our manufacturing process ensures that each batch meets rigorous international standards for purity and consistency. As a dedicated chemical supplier, we understand the importance of reliable raw materials in drug development and large-scale production.
The chemical structure incorporates a benzyloxycarbonyl-protected valine moiety linked to a ganciclovir derivative. This specific configuration allows for selective reactivity during subsequent synthetic steps. Our facility employs state-of-the-art organic synthesis techniques to maintain the structural integrity of the molecule. Clients in the pharmaceutical sector rely on this intermediate for developing next-generation therapeutic solutions.
Key Specifications
| Parameter | Specification |
|---|---|
| Product Name | CBZ-Valganciclovir |
| CAS Number | 194154-40-0 |
| Molecular Formula | C22H28N6O7 |
| Molecular Weight | 488.49 g/mol |
| Appearance | White to off-white powder |
| Purity (GC) | ≥98.0% |
| Storage Condition | -20°C Freezer |
Quality Assurance
Quality control is paramount in the production of pharmaceutical intermediates. Every shipment of CBZ-Valganciclovir undergoes comprehensive analytical testing. We utilize gas chromatography and other advanced methods to verify assay levels. Our standard test results consistently show purity levels exceeding 98.5%, ensuring optimal performance in downstream reactions. A Certificate of Analysis (COA) is provided with every order to guarantee transparency and compliance.
Industrial Applications
- Pharmaceutical synthesis for antiviral medications
- Research and development of nucleoside analogues
- Custom organic synthesis projects
- Production of active pharmaceutical ingredients (APIs)
Storage and Handling
To maintain stability and potency, this product should be stored in a sealed container at -20°C. Proper handling procedures must be followed to prevent contamination or degradation. We recommend consulting the safety data sheet (SDS) for detailed handling instructions. Our logistics team ensures cold-chain shipping options are available for global delivery. Contact our technical support team for specific storage recommendations based on your facility requirements.



