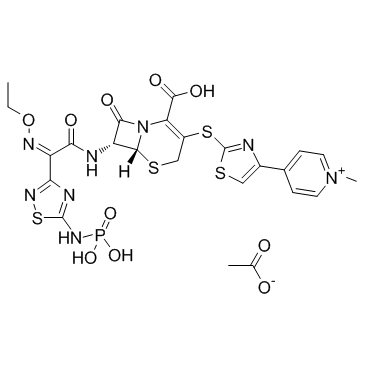
Ceftaroline Fosamil Acetate
- CAS No.400827-46-5
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
Premium grade Ceftaroline Fosamil Acetate for pharmaceutical manufacturing. High purity antibiotic intermediate compliant with global standards.
Request Bulk PricingProduct Technical Details
Product Overview
Ceftaroline Fosamil Acetate represents a critical advancement in modern pharmaceutical chemistry, specifically designed to address the growing global challenge of antibiotic resistance. As a fifth-generation cephalosporin prodrug, this compound serves as a vital active pharmaceutical ingredient intermediate for the development of injectable solutions targeting serious bacterial infections. Our manufacturing process ensures exceptional purity and structural integrity, making it an ideal choice for pharmaceutical companies seeking reliable supply chains for anti-infective therapies.
The chemical structure incorporates a phosphonoamino group which significantly enhances aqueous solubility, allowing for effective intravenous administration. Upon administration, the fosamil moiety is rapidly cleaved by phosphatases to release the bioactive ceftaroline. This sophisticated design underscores the importance of high-quality synthesis during the intermediate stage. We employ rigorous analytical methods to verify every batch, ensuring that the complex bicyclic core remains stable and free from detrimental impurities that could affect efficacy or safety profiles.
Key Specifications
Quality assurance is the cornerstone of our production facility. Each lot undergoes comprehensive testing using high-performance liquid chromatography and nuclear magnetic resonance spectroscopy. These techniques confirm not only the chemical identity but also the precise quantification of related substances and residual solvents. The acetate salt form provides additional stability during storage and transport, crucial for maintaining potency in global distribution networks. Our commitment to Good Manufacturing Practices guidelines ensures consistency across large-scale production runs.
| Item | Specifications |
|---|---|
| Appearance | White to light yellow solid |
| Structural Identification | Conforms to standard spectrum (1H NMR) |
| Related Substance U3 | NMT 0.80% |
| Purity (HPLC) | NLT 99.0% |
| Acetic Acid Content | 7.1% to 8.7% |
| Water Content | NMT 3.0% |
Industrial Applications
In the context of contemporary medicinal chemistry, the demand for robust anti-MRSA agents has never been higher. Ceftaroline Fosamil Acetate fills this niche by offering broad-spectrum activity against Gram-positive pathogens, including strains resistant to methicillin. For pharmaceutical developers, securing a stable source of this intermediate is strategic. Our facility is equipped to handle complex organic synthesis involving sensitive functional groups such as the thiazolyl sulfanyl moiety and the ethoxyimino side chain. Protecting these groups during synthesis requires specialized expertise and state-of-the-art reactor systems.
Key technical advantages include superior control over stereocenters at the 6R and 7R positions, which are essential for biological activity. The manufacturing route is optimized to minimize environmental impact while maximizing yield. We understand that downstream formulation partners require materials with predictable physical properties. Therefore, we monitor particle size distribution and moisture content meticulously. This attention to detail facilitates smoother processing during the final drug product manufacturing stage, reducing downtime and ensuring batch-to-batch reproducibility.
Quality Assurance and Supply
Furthermore, regulatory compliance is paramount. We provide full documentation support, including Certificates of Analysis, Method Validation Reports, and Stability Data. This transparency accelerates the regulatory filing process for our clients. The specification profile typically includes strict limits on related substances, ensuring that impurities are kept well below threshold values. Acetic acid content is controlled to maintain the correct stoichiometry of the salt form, while water content is minimized to prevent hydrolysis during storage.
Our logistics network supports global delivery, ensuring that temperature-sensitive materials are handled according to recommended storage conditions. Whether for clinical trial supplies or commercial scale manufacturing, our team collaborates closely with clients to meet specific timeline requirements. We invite partners to request samples for evaluation and discuss custom packaging options tailored to their production lines. By choosing our Ceftaroline Fosamil Acetate, manufacturers gain a partner dedicated to quality, reliability, and scientific excellence in the fight against resistant bacterial infections.



