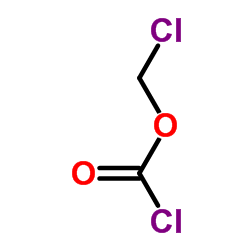
Chloromethyl Carbonochloridate
- CAS No.22128-62-7
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Chloromethyl Carbonochloridate (CAS 22128-62-7) designed for pharmaceutical synthesis. Reliable supply for global API manufacturing.
Request Bulk PricingProduct Technical Details
Product Overview
Chloromethyl Carbonochloridate, also widely recognized in the industry as Chloromethyl chloroformate, is a critical reactive intermediate utilized extensively in organic synthesis and pharmaceutical manufacturing. With the CAS registry number 22128-62-7, this chemical compound serves as a vital building block for the production of various active pharmaceutical ingredients. Our facility specializes in the large-scale production of this reagent, ensuring consistent quality and supply chain stability for global partners.
As a chloroformate derivative, this substance exhibits high reactivity, making it indispensable for introducing chloromethyl carbonyl groups into complex molecular structures. The manufacturing process adheres to strict international safety and quality standards, guaranteeing that each batch meets the rigorous demands of modern drug synthesis.
Technical Specifications
| Parameter | Specification |
|---|---|
| Chemical Name | Chloromethyl Carbonochloridate |
| CAS Number | 22128-62-7 |
| Molecular Formula | C2H2Cl2O2 |
| Molecular Weight | 128.94 g/mol |
| Appearance | Colorless liquid |
| Purity | ≥98.0% |
| Density | 1.449 g/mL at 20 °C |
| Boiling Point | 107-108 °C |
| Flash Point | 95 °C |
| Refractive Index | 1.428 |
Industrial Applications
This intermediate is primarily employed in the synthesis of sophisticated pharmaceutical compounds. It acts as a key precursor in the manufacturing pathways for several important therapeutic agents. Notable applications include its role as an intermediate in the synthesis of Metronidazole, a widely used antibiotic and antiprotozoal medication. Furthermore, it is integral to the production processes of Tenofovir and Tenofovir disoproxil fumarate, which are essential components in antiretroviral therapy.
- Pharmaceutical API Synthesis
- Organic Chemical Intermediates
- Fine Chemical Manufacturing
- Research and Development Reagent
Quality Assurance and Safety
Quality control is paramount in our production facility. Every batch of Chloromethyl Carbonochloridate undergoes comprehensive testing using advanced analytical techniques such as Gas Chromatography (GC) and High-Performance Liquid Chromatography (HPLC). We provide a Certificate of Analysis (COA) with every shipment to verify purity and physical properties. Due to the reactive nature of chloroformates, strict safety protocols are enforced during handling. The compound must be protected from moisture and strong light to maintain stability.
Packaging and Storage
To ensure product integrity during transit, we offer flexible packaging solutions tailored to customer requirements. Standard packing includes 25kg cardboard drums, though custom configurations are available upon request. Storage recommendations dictate keeping the container tightly closed in a cool, dry, and well-ventilated area. It is crucial to keep the material away from moisture, strong heat, and direct sunlight to prevent degradation. Our logistics team ensures compliant shipping practices for hazardous chemicals, facilitating smooth global delivery.



