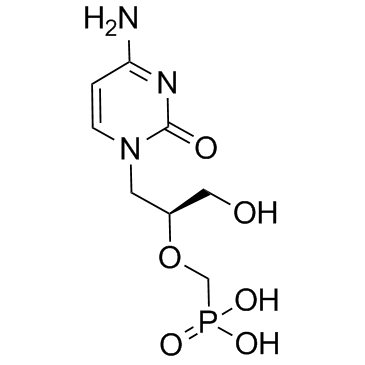
Cidofovir Anhydrous
- CAS No.113852-37-2
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Cidofovir Anhydrous for pharmaceutical manufacturing. Reliable supply with COA.
Request Bulk PricingProduct Technical Details
Product Overview
Cidofovir Anhydrous is a high-quality pharmaceutical intermediate designed for professional research and manufacturing applications. As a nucleotide analogue, this compound plays a critical role in the development of advanced antiviral therapeutics. Our facility ensures strict quality control measures are implemented at every stage of production to guarantee consistency and purity suitable for demanding pharmaceutical synthesis processes.
We understand the importance of reliability in the supply chain for active pharmaceutical ingredients. Therefore, our Cidofovir Anhydrous is manufactured under rigorous standards to meet global regulatory requirements for chemical intermediates. This product is intended for use by licensed pharmaceutical manufacturers and research institutions engaged in the development of antiviral medications.
Technical Specifications
| Parameter | Specification |
|---|---|
| Product Name | Cidofovir Anhydrous |
| CAS Number | 113852-37-2 |
| Molecular Formula | C8H14N3O6P |
| Molecular Weight | 279.19 g/mol |
| Purity | >99% |
| Appearance | Soft White Powder |
| Density | 1.76 g/cm3 |
| Melting Point | 260°C |
| Storage Conditions | Store in a cool, dry place |
Industrial Applications
This chemical intermediate is primarily utilized in the pharmaceutical industry for the synthesis of antiviral agents. It serves as a key building block in the formulation of medications targeting specific viral replication mechanisms. Due to its high stability and reactivity profile, it is an ideal candidate for large-scale production environments where consistency is paramount.
Researchers and formulation scientists value this compound for its well-defined physicochemical properties. It is commonly employed in projects focused on developing treatments for serious viral infections. Our supply supports the creation of injectable formulations and other specialized delivery systems required for complex therapeutic protocols.
Quality Assurance and Handling
Quality is the cornerstone of our manufacturing process. Each batch of Cidofovir Anhydrous undergoes comprehensive analytical testing to verify identity, purity, and potency. We provide a Certificate of Analysis (COA) with every shipment to ensure transparency and compliance with your internal quality standards.
- Strict adherence to Good Manufacturing Practices (GMP) guidelines.
- Advanced analytical instrumentation for precise quality verification.
- Secure packaging to maintain stability during international transit.
- Full traceability from raw materials to finished product.
Proper handling procedures should be followed to maintain the integrity of the material. Personnel should utilize appropriate personal protective equipment when handling this substance. Storage should be maintained in a controlled environment to prevent degradation due to moisture or excessive heat. Our logistics team ensures that all shipments are packaged securely to arrive in optimal condition.
Why Choose Our Supply
As a global manufacturer, we offer competitive bulk pricing without compromising on quality. Our extensive experience in the chemical industry allows us to provide technical support and customization options for large-scale projects. We are committed to fostering long-term partnerships with clients who require dependable sources of high-performance chemical intermediates.
Contact our sales team today to discuss your specific requirements. We are ready to assist with sample requests, technical documentation, and logistics planning to streamline your procurement process. Trust us to be your partner in advancing pharmaceutical innovation through superior chemical supply solutions.



