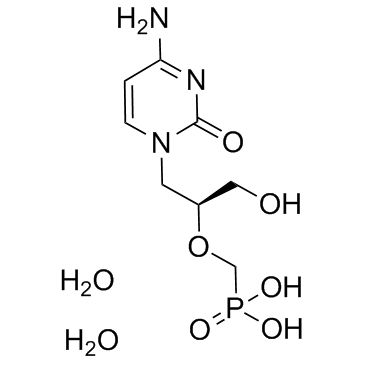
Cidofovir Dihydrate
- CAS No.149394-66-1
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Cidofovir Dihydrate (CAS 149394-66-1) supplied for pharmaceutical research and development. Available in bulk quantities with full documentation.
Request Bulk PricingProduct Technical Details
Product Overview
Cidofovir Dihydrate is a high-value pharmaceutical intermediate characterized by its specific nucleotide analog structure. This compound serves as a critical building block in the development of antiviral therapeutics. Our manufacturing process ensures exceptional consistency and purity, making it suitable for rigorous scientific research and large-scale pharmaceutical production. As a leading global supplier, we prioritize quality assurance at every stage of synthesis and packaging.
The chemical integrity of Cidofovir Dihydrate is maintained through advanced crystallization techniques that stabilize the hydrate form. This stability is essential for maintaining the compound's reactivity and performance during downstream formulation processes. We adhere to strict international standards to deliver a product that meets the demanding requirements of modern medicinal chemistry.
Technical Specifications
| Parameter | Specification |
|---|---|
| Product Name | Cidofovir Dihydrate |
| CAS Number | 149394-66-1 |
| Molecular Formula | C8H14N3O6P |
| Molecular Weight | 279.19 (Anhydrous basis) |
| Purity | >99% |
| Appearance | White Powder |
| Melting Point | 260°C (dec) |
| Density | 1.76 g/cm3 |
Quality Assurance And Manufacturing
Our production facilities operate under stringent quality control protocols to guarantee the reliability of every batch. Each lot of Cidofovir Dihydrate undergoes comprehensive analytical testing, including High-Performance Liquid Chromatography (HPLC) and Nuclear Magnetic Resonance (NMR) spectroscopy. These tests verify chemical identity, assay potency, and impurity profiles to ensure compliance with specified standards.
We provide a complete Certificate of Analysis (COA) with every shipment, detailing all critical quality attributes. Our supply chain is optimized for global distribution, ensuring timely delivery while maintaining product stability during transit. Customers can rely on our technical support team for assistance with handling, storage, and integration into their specific research workflows.
Applications And Research Use
This compound is primarily utilized in the pharmaceutical industry as an active pharmaceutical ingredient (API) precursor or intermediate. It plays a vital role in the synthesis of antiviral agents targeting DNA polymerase enzymes. Researchers utilize this material for preclinical studies, formulation development, and bioequivalence assessments.
- Ideal for antiviral drug discovery and development programs.
- Suitable for use in controlled laboratory environments for analytical standard preparation.
- Supports large-scale manufacturing of injectable pharmaceutical formulations.
- Compatible with various organic synthesis routes requiring high-purity nucleotide analogs.
Storage And Handling
To preserve the chemical stability of Cidofovir Dihydrate, proper storage conditions are essential. The material should be kept in a cool, dry place away from direct sunlight and moisture. Containers must be tightly sealed to prevent hydration changes or contamination. Personnel handling this compound should follow standard laboratory safety procedures, including the use of appropriate personal protective equipment.



