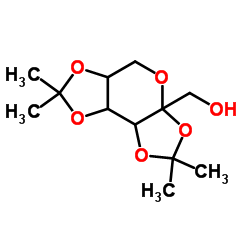
Diacetonefructose
- CAS No.20880-92-6
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity Diacetonefructose (CAS 20880-92-6) serving as a critical carbohydrate intermediate for pharmaceutical synthesis. Available in bulk quantities with comprehensive COA.
Request Bulk PricingProduct Technical Details
Product Overview
Diacetonefructose, chemically known as 2,3:4,5-di-O-isopropylidenefructose, is a specialized carbohydrate derivative widely utilized in the field of organic synthesis and pharmaceutical manufacturing. As a protected form of D-fructose, this compound features isopropylidene groups that safeguard specific hydroxyl functionalities, enabling selective chemical transformations in complex molecular architectures. Our facility produces this intermediate under strict quality control protocols to ensure consistency and reliability for downstream applications.
This material is particularly valued in the synthesis of nucleoside analogs, glycosides, and various fine chemicals where precise stereochemistry is paramount. By stabilizing the fructofuranose or fructopyranose ring structure, it serves as a robust building block for researchers and industrial chemists aiming to develop advanced therapeutic agents or functional materials.
Chemical Characteristics
The molecular structure of Diacetonefructose (C12H20O6) provides unique reactivity profiles essential for multi-step synthesis routes. The dual acetonide protection allows chemists to perform reactions at unprotected sites without affecting the protected regions, thereby streamlining synthetic pathways. This selectivity reduces waste and improves overall yield in manufacturing processes.
Physically, the compound presents as white needle-like crystals with a defined melting point range, ensuring easy handling and processing in standard laboratory and plant environments. Its stability under recommended storage conditions makes it a dependable choice for long-term projects requiring consistent raw material performance.
Industrial Applications
Primarily categorized as a pharmaceutical intermediate, this compound plays a vital role in the production of active pharmaceutical ingredients (APIs). It is frequently referenced in analytical contexts, such as being identified as Topiramate Related Compound A in quality control specifications for certain anticonvulsant medications. However, our supply is strictly intended for chemical synthesis and research purposes.
- Utilized in the synthesis of complex carbohydrate derivatives.
- Serves as a chiral pool starting material for asymmetric synthesis.
- Employed in the development of nucleoside analogues for antiviral research.
- Acts as a reference standard for analytical method validation.
Quality Specifications
We adhere to rigorous manufacturing standards to guarantee high purity and low impurity profiles. Each batch undergoes comprehensive testing including HPLC, IR spectroscopy, and physical constant verification. The following table outlines typical specification parameters and test results from our recent production runs.
| Items of Analysis | Standard of Analysis | Test Results |
|---|---|---|
| Appearance | White to off-white powder | White powder |
| IR | IR spectrum match with reference standard | Conforms |
| Water (KF) | ≤0.5% | 0.3% |
| Residue on Ignition | ≤0.5% | 0.4% |
| Optical rotation | -34.8°~-30.5° | -32.6° |
| Loss on drying | ≤1.0% | 0.4% |
| Heavy metals | ≤20ppm | Conforms |
| Melting Point | 94.0-97.5°C | 94.4-94.7°C |
| Assay (HPLC) | 98.0%~102.0% | 99.0% |
| Conclusion | Conforms to Factory Standard | |
Storage and Handling
To maintain optimal quality, Diacetonefructose should be stored in a cool, dry environment away from direct sunlight and moisture. The recommended storage temperature is below -20°C for extended periods, though short-term storage at controlled room temperature is acceptable if sealed properly. Always utilize appropriate personal protective equipment when handling chemical intermediates in a laboratory setting.
Our global logistics network ensures secure packaging and timely delivery. We provide full documentation including Certificates of Analysis (COA), Safety Data Sheets (SDS), and method validation reports to support your regulatory compliance needs. For bulk pricing and technical inquiries, please contact our sales team directly.



