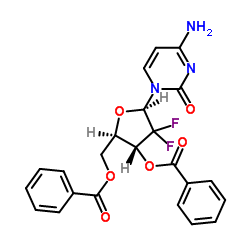
2',2'-Difluoro-2'-deoxycytidine-3',5'-dibenzoate
- CAS No.134790-39-9
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity pharmaceutical intermediate essential for Gemcitabine API synthesis, offering consistent quality and reliable bulk supply for global manufacturers.
Request Bulk PricingProduct Technical Details
Product Overview
2',2'-Difluoro-2'-deoxycytidine-3',5'-dibenzoate serves as a critical advanced intermediate in the pharmaceutical industry, specifically designed for the synthesis of Gemcitabine, a widely recognized antineoplastic agent. This compound features a specialized nucleoside structure with difluoro substitution, requiring precise chemical engineering to maintain stability and reactivity during downstream processing. Our manufacturing facility adheres to strict quality management systems to ensure every batch meets the rigorous demands of API production.
As a key building block, this dibenzoate derivative protects the hydroxyl groups during specific synthetic steps, allowing for selective reactions elsewhere on the molecule. The high level of purity achieved in our production process minimizes impurity carryover into the final active pharmaceutical ingredient, thereby supporting regulatory compliance and patient safety. We understand the complexities of nucleoside chemistry and have optimized our synthesis routes to maximize yield while maintaining exceptional chemical integrity.
Technical Specifications
Quality control is paramount in the production of pharmaceutical intermediates. Each lot undergoes comprehensive analytical testing using validated methods including HPLC, NMR, and mass spectrometry. The physical properties are tightly controlled to ensure consistent performance in subsequent reaction steps. Below are the standard specifications for this material:
| Parameter | Specification |
|---|---|
| CAS Number | 134790-39-9 |
| Molecular Formula | C23H19F2N3O6 |
| Molecular Weight | 471.41 g/mol |
| Appearance | Off-white solid |
| Purity | ≥98.0% |
| Melting Point | 229-232ºC |
| Density | 1.459 g/cm3 |
Industrial Applications
This intermediate is primarily utilized in the multi-step synthesis of Gemcitabine hydrochloride. The dibenzoate protection strategy is crucial for managing stereochemistry and preventing unwanted side reactions during the formation of the glycosidic bond. Pharmaceutical manufacturers rely on consistent supply chains for such specialized chemicals to maintain their own production schedules. Our facility supports scale-up from pilot batches to commercial tonnage, ensuring continuity for long-term projects.
- Essential precursor for antineoplastic API manufacturing
- Optimized for high-yield coupling reactions
- Compatible with standard deprotection protocols
- Supports GMP compliant downstream processing
- Reduced impurity profile for easier purification
Quality Assurance and Handling
We implement a robust quality assurance program that covers raw material sourcing, in-process control, and final release testing. Documentation such as Certificates of Analysis (COA) and Method Validation Reports are provided with every shipment. Proper handling is required to maintain product stability. The material should be stored in a cool, ventilated environment away from direct sunlight and moisture. Packaging is designed to prevent contamination during transit, with options available for drums or customized containers based on client logistics requirements. Our technical team is available to support process optimization and regulatory documentation needs.



