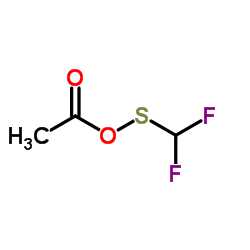
Difluoromethylthioacetic Acid
- CAS No.83494-32-0
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Difluoromethylthioacetic Acid (CAS 83494-32-0) designed for pharmaceutical synthesis. Ideal intermediate for Flomoxef production with ≥99.0% purity assurance.
Request Bulk PricingProduct Technical Details
Product Overview
Difluoromethylthioacetic Acid is a specialized fluorinated organic compound that serves as a critical building block in modern pharmaceutical chemistry. Characterized by its unique difluoromethylthio functional group, this chemical exhibits distinct reactivity profiles that are essential for constructing complex molecular architectures. Our manufacturing process ensures consistent quality and high purity levels, making it a preferred choice for research laboratories and industrial synthesis facilities focused on advanced antibiotic development.
As a key precursor in the synthesis of Flomoxef, this intermediate plays a vital role in the production of next-generation therapeutic agents. The incorporation of fluorine atoms enhances the metabolic stability and bioavailability of the final drug product, underscoring the importance of using high-grade raw materials during the early stages of synthesis. We adhere to strict quality control protocols to guarantee that every batch meets rigorous international standards.
Technical Specifications
| Parameter | Value |
|---|---|
| CAS Number | 83494-32-0 |
| Molecular Formula | C3H4F2O2S |
| Molecular Weight | 142.12 g/mol |
| Appearance | Off-white to white crystalline powder |
| Purity | ≥99.0% |
| Density | 1.255 g/cm³ |
| Boiling Point | 126.2°C |
| Flash Point | 30.1°C |
| Refractive Index | 1.396 |
Industrial Applications
The primary application of Difluoromethylthioacetic Acid lies within the pharmaceutical sector, specifically as an intermediate for the synthesis of Flomoxef, a cephamycin antibiotic. Beyond this specific use case, the compound is valuable in organic synthesis where fluorinated sulfur-containing motifs are required. Chemists utilize this building block to introduce specific electronic and steric properties into target molecules, facilitating the development of novel agrochemicals and medicinal compounds.
Our supply chain is optimized to support large-scale manufacturing needs. Whether for pilot plant operations or full commercial production, we provide the consistency required for reproducible results. The chemical's stability under standard storage conditions allows for flexible logistics planning, ensuring that production schedules are maintained without interruption due to material degradation.
Quality Assurance and Storage
We understand that purity is paramount in pharmaceutical intermediate synthesis. Each production batch undergoes comprehensive analytical testing, including HPLC and NMR spectroscopy, to verify identity and purity levels. A Certificate of Analysis (COA) is provided with every shipment, documenting all critical quality attributes. Our facility operates under stringent safety and environmental guidelines to minimize risk and ensure product integrity.
- Stored in a cool, ventilated place away from direct sunlight.
- Packaged in 25 kg drums or customized according to client requirements.
- Compatible with standard hazardous material shipping protocols.
- Long shelf life when stored under recommended conditions.
For technical inquiries or to request a sample for evaluation, please contact our sales team. We are committed to supporting your synthesis projects with reliable materials and expert service.



