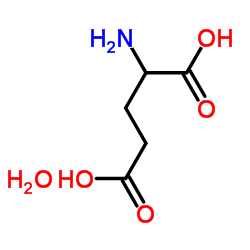
DL-Glutamic Acid Monohydrate
- CAS No.19285-83-7
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity DL-Glutamic Acid Monohydrate designed for pharmaceutical synthesis and industrial applications with consistent quality assurance.
Request Bulk PricingProduct Technical Details
Product Overview
DL-Glutamic Acid Monohydrate represents a critical component in the landscape of modern pharmaceutical and chemical synthesis. As a stable hydrated form of the amino acid glutamic acid, this compound offers superior handling characteristics for industrial-scale processes. Our manufacturing facility ensures that every batch meets rigorous international standards, providing a reliable foundation for downstream chemical production. The monohydrate structure enhances stability during storage and transport, making it an ideal choice for complex organic synthesis pathways.
This chemical substance is widely recognized for its versatility in creating various amino acid derivatives. Whether utilized in laboratory research or large-scale manufacturing, the consistency of our product allows for predictable reaction outcomes. We prioritize purity and structural integrity, ensuring that the material performs optimally in sensitive synthetic environments.
Technical Specifications
Quality control is paramount in the production of fine chemical intermediates. Our DL-Glutamic Acid Monohydrate undergoes comprehensive testing to verify compliance with specified parameters. The following table outlines the key physical and chemical properties that define our standard grade material.
| Parameter | Specification |
|---|---|
| CAS Number | 19285-83-7 |
| Molecular Formula | C5H11NO5 |
| Molecular Weight | 165.145 |
| Appearance | White powder |
| Assay | ≥98.0% |
| Melting Point | 194 °C |
| Loss on Drying | ≤0.50% |
| Residue on Ignition | ≤0.20% |
| Heavy Metals | ≤10ppm |
| Chloride | ≤0.10% |
Industrial Applications
Primarily utilized as a pharmaceutical intermediate, this compound serves as a foundational building block for numerous synthetic routes. Its chemical structure allows for diverse functionalization, enabling chemists to develop advanced therapeutic agents and specialized organic compounds. The high purity level ensures minimal interference in catalytic processes, reducing the need for extensive purification steps downstream.
Beyond pharmaceuticals, this amino acid derivative finds utility in various chemical manufacturing sectors. It is frequently employed in the production of specialty chemicals where precise stoichiometry is required. The reliability of the raw material directly impacts the efficiency of the final production line, making supplier consistency a key factor for procurement managers.
Quality Assurance and Packaging
We maintain a robust quality management system to guarantee product integrity from synthesis to delivery. Each shipment is accompanied by a Certificate of Analysis (COA) detailing batch-specific test results. Our packaging solutions are designed to protect the material from moisture and contamination during transit. Standard packaging includes 25kg drums, though we offer flexible customization to meet specific logistical requirements.
Proper storage is essential to maintain the physical properties of the monohydrate form. We recommend storing the product in a cool, dry, and well-ventilated area. Containers should remain tightly closed when not in use to prevent absorption of atmospheric moisture. By adhering to these guidelines, customers can ensure the long-term stability and performance of the chemical in their respective applications.



