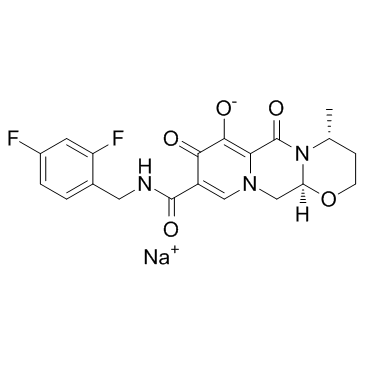
Dolutegravir Sodium
- CAS No.1051375-19-9
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity Dolutegravir Sodium API intermediate for pharmaceutical manufacturing. Available in bulk quantities with comprehensive COA.
Request Bulk PricingProduct Technical Details
Product Overview
Dolutegravir Sodium represents a high-value pharmaceutical intermediate widely utilized in the development and production of advanced antiretroviral therapies. As a sodium salt form of the active compound, this material is engineered to meet stringent quality standards required by modern pharmaceutical manufacturing processes. Our facility specializes in the supply of this critical chemical entity, ensuring consistent batch-to-batch reliability for research institutions and production partners globally.
This compound is characterized by its specific molecular structure designed for stability and solubility in various formulation environments. We prioritize the delivery of materials that adhere to rigorous industrial purity specifications, supporting the seamless integration of this intermediate into downstream synthetic routes. Our commitment to quality ensures that every shipment meets the detailed requirements necessary for complex pharmaceutical applications.
Technical Specifications
| Parameter | Specification |
|---|---|
| Chemical Name | Dolutegravir Sodium |
| CAS Registry Number | 1051375-19-9 |
| Molecular Formula | C20H18F2N3NaO5 |
| Molecular Weight | 441.361 g/mol |
| Appearance | White to light yellow powder |
| Purity Assay | ≥98% |
| Packaging | 25 kg/drum or customized |
| Storage Conditions | Cool, ventilated area |
Industrial Applications
In the context of pharmaceutical development, this substance serves as a crucial building block for therapeutic formulations. It is primarily employed in the synthesis of medications targeting specific viral mechanisms within controlled research environments. The chemical properties of this sodium salt facilitate efficient processing during tablet manufacturing and other dosage form preparations. Manufacturers rely on this intermediate for its consistent performance profile and compatibility with standard excipients.
Our supply chain is optimized to support large-scale production needs while maintaining the integrity of the chemical structure. We understand the critical nature of supply continuity in the pharmaceutical sector and have established robust logistics to ensure timely delivery. Whether for pilot-scale studies or full commercial production, our material provides the foundational quality required for successful outcomes.
Quality Assurance and Handling
Every batch of Dolutegravir Sodium undergoes comprehensive analytical testing to verify identity, purity, and physical characteristics. We provide a Certificate of Analysis (COA) with each shipment, documenting compliance with agreed-upon specifications. Proper handling procedures are recommended to maintain stability, including storage in a cool and well-ventilated environment away from direct moisture and light exposure.
- Strict quality control protocols at every manufacturing stage
- Comprehensive documentation including COA and MSDS
- Flexible packaging solutions to meet specific client requirements
- Global shipping capabilities with secure containment
- Dedicated technical support for integration queries
Partnering with us ensures access to a reliable source of high-performance pharmaceutical intermediates. We are dedicated to supporting the advancement of medical research and production through the supply of premium chemical materials that meet the evolving demands of the industry.



