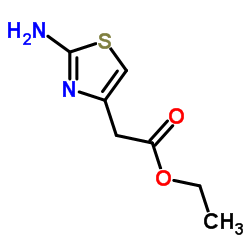
Ethyl 2-Amino-4-Thiazoleacetate
- CAS No.53266-94-7
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Ethyl 2-Amino-4-Thiazoleacetate (CAS 53266-94-7) designed for efficient cephalosporin antibiotic synthesis. Reliable supply for pharmaceutical manufacturing.
Request Bulk PricingProduct Technical Details
Product Overview
Ethyl 2-Amino-4-Thiazoleacetate is a critical pharmaceutical intermediate widely utilized in the synthesis of advanced cephalosporin antibiotics. As a key thiazole derivative, this compound plays an indispensable role in constructing the side chains required for modern beta-lactam medications. Our manufacturing process ensures exceptional chemical stability and high purity levels, making it an ideal choice for large-scale pharmaceutical production.
This esterified thiazole compound is characterized by its robust molecular structure, which facilitates efficient coupling reactions during antibiotic synthesis. The presence of the amino group at the second position and the ester functionality allows for versatile chemical modifications, enabling medicinal chemists to develop potent therapeutic agents with improved pharmacokinetic profiles.
Key Specifications
| Parameter | Value |
|---|---|
| CAS Number | 53266-94-7 |
| Molecular Formula | C7H10N2O2S |
| Molecular Weight | 186.23 g/mol |
| Purity | ≥99.0% |
| Melting Point | 92-94°C |
| Appearance | White or creamy white crystalline powder |
| Density | 1.295 g/cm³ |
| Boiling Point | 318.5°C |
Industrial Applications
The primary application of Ethyl 2-Amino-4-Thiazoleacetate lies in the production of cefotiam and related cephalosporin antibiotics. These antibiotics are essential for treating a wide range of bacterial infections, making the supply chain for this intermediate vital for global public health. Our product is specifically optimized to meet the stringent requirements of GMP-certified pharmaceutical facilities.
Beyond antibiotic synthesis, this thiazole building block is increasingly explored in medicinal chemistry research for developing novel therapeutic agents. Its unique heterocyclic structure provides a valuable scaffold for drug discovery programs focused on infectious diseases and other therapeutic areas.
Quality Assurance and Storage
We maintain rigorous quality control protocols throughout the manufacturing process. Each batch undergoes comprehensive testing using HPLC and NMR spectroscopy to verify identity and purity. A Certificate of Analysis (COA) is provided with every shipment to ensure full traceability and compliance with international standards.
- Packaged in 25 kg drums or according to specific customer requirements.
- Stored in a cool, ventilated environment to maintain stability.
- Protected from moisture and direct sunlight during transportation.
- Global shipping capabilities with secure logistics partners.
Our commitment to quality extends beyond the product itself. We offer technical support to assist clients with integration into their synthesis routes, ensuring optimal yields and process efficiency. For bulk pricing inquiries or custom packaging solutions, please contact our sales team for detailed quotations and lead time information.



