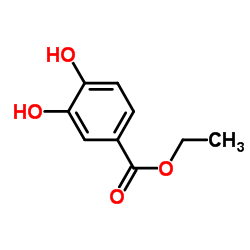
Ethyl 3,4-dihydroxybenzoate
- CAS No.3943-89-3
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Ethyl 3,4-dihydroxybenzoate (CAS 3943-89-3) designed for pharmaceutical synthesis. Reliable supply for Erlotinib intermediate production with strict quality control.
Request Bulk PricingProduct Technical Details
Product Overview
Ethyl 3,4-dihydroxybenzoate, frequently recognized in the industry as Ethyl Protocatechuate, stands as a vital organic compound within the pharmaceutical and fine chemical sectors. This ester derivative of protocatechuic acid is characterized by its distinct chemical stability and reactivity, making it an indispensable building block for complex molecular synthesis. Our facility specializes in the large-scale production of this intermediate, ensuring consistent quality that meets the rigorous demands of modern drug development pipelines.
As a key precursor, this compound plays a significant role in the construction of advanced therapeutic agents. The molecular structure, featuring a dihydroxy benzene ring coupled with an ethyl ester group, provides versatile reaction sites for further functionalization. We maintain strict control over every batch to guarantee that the chemical integrity remains intact from synthesis to final packaging.
Technical Specifications
Quality assurance is the cornerstone of our manufacturing process. Each production lot undergoes comprehensive analytical testing to verify compliance with international standards. The following table outlines the critical physical and chemical parameters associated with our premium grade material.
| Parameter | Specification |
|---|---|
| CAS Number | 3943-89-3 |
| Molecular Formula | C9H10O4 |
| Molecular Weight | 182.17 g/mol |
| Appearance | White crystalline powder |
| Purity | ≥99.0% |
| Melting Point | 132-134 °C |
| Loss on Drying | ≤1.0% |
| Boiling Point | 358.1 °C at 760 mmHg |
Industrial Applications
The primary application of Ethyl 3,4-dihydroxybenzoate lies in the synthesis of sophisticated pharmaceutical intermediates. It is notably utilized in the manufacturing process of kinase inhibitors, such as Erlotinib hydrochloride. The high purity levels we provide are essential for ensuring the efficacy and safety of the final active pharmaceutical ingredients (APIs). Beyond oncology research, this chemical serves as a valuable reagent in organic synthesis laboratories focusing on heterocyclic compounds and antioxidant derivatives.
Our clients leverage this material for various research and development projects where structural precision is paramount. The consistent batch-to-batch reproducibility allows process chemists to optimize reaction conditions without variability concerns. Whether used in pilot-scale trials or commercial production, our product supports seamless integration into existing synthetic routes.
Quality Assurance and Packaging
We understand that contamination control is critical in pharmaceutical manufacturing. Therefore, our production environment adheres to strict hygiene and safety protocols. Every shipment is accompanied by a comprehensive Certificate of Analysis (COA) detailing the test results for identity, purity, and residual solvents. This documentation ensures full traceability and regulatory compliance for our global partners.
Standard packaging consists of 25kg cardboard drums, designed to protect the material from moisture and physical damage during transit. Custom packaging solutions are available upon request to accommodate specific logistical requirements. Proper storage involves keeping the containers in a cool, dry, and well-ventilated area, away from strong light and heat sources to maintain optimal stability over extended periods.



