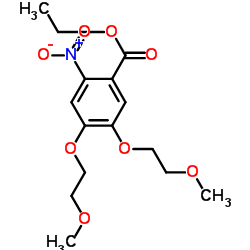
Ethyl 4,5-bis(2-methoxyethoxy)-2-nitrobenzoate
- CAS No.179688-26-7
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Ethyl 4,5-bis(2-methoxyethoxy)-2-nitrobenzoate (CAS 179688-26-7) designed for pharmaceutical synthesis. Reliable supply for Erlotinib intermediate production with strict quality control.
Request Bulk PricingProduct Technical Details
Product Overview
Ethyl 4,5-bis(2-methoxyethoxy)-2-nitrobenzoate is a specialized pharmaceutical intermediate characterized by its high chemical stability and precise molecular structure. Identified by CAS number 179688-26-7, this compound serves as a critical building block in the synthesis of advanced therapeutic agents. Our manufacturing process ensures consistent quality, making it an ideal choice for research and development teams focused on kinase inhibitor production.
As a key precursor in the pharmaceutical industry, this nitrobenzoate derivative undergoes rigorous testing to meet international standards. The presence of methoxyethoxy groups enhances its solubility profile and reactivity during subsequent synthetic steps. We prioritize purity and safety in every batch produced, supporting global supply chains for essential medications.
Technical Specifications
| Parameter | Value |
|---|---|
| Chemical Name | Ethyl 4,5-bis(2-methoxyethoxy)-2-nitrobenzoate |
| CAS Number | 179688-26-7 |
| Molecular Formula | C15H21NO8 |
| Molecular Weight | 343.33 g/mol |
| Purity | ≥98% |
| Appearance | White or off white powder |
| Density | 1.215 g/cm3 |
| Boiling Point | 467.8°C at 760 mmHg |
| Flash Point | 185.6°C |
Industrial Applications
This compound is primarily utilized as an intermediate in the synthesis of Erlotinib hydrochloride, a well-known pharmaceutical agent. The high purity level of ≥98% ensures minimal impurity carryover into the final active pharmaceutical ingredient (API). Manufacturers rely on this material for its consistent performance in multi-step organic synthesis routes.
- Essential building block for Erlotinib production
- Suitable for large-scale pharmaceutical manufacturing
- Compatible with standard organic synthesis protocols
- Supports development of targeted cancer therapies
Quality Assurance and Storage
Quality control is paramount in our production facility. Each batch is accompanied by a Certificate of Analysis (COA) verifying identity, purity, and physical properties. We employ advanced analytical techniques such as HPLC and NMR to confirm structural integrity. Moisture content is strictly controlled to prevent degradation during storage.
For optimal stability, store this material in a cool and dry well-closed container. Keep away from moisture and strong light or heat sources. Proper handling ensures the longevity of the chemical properties required for sensitive synthetic applications. Our packaging options include 25kg cardboard drums or custom specifications based on client requirements.
Global Supply and Support
We operate as a global manufacturer committed to delivering reliable chemical solutions. Our logistics network ensures timely delivery while maintaining product integrity during transit. Clients benefit from competitive bulk pricing and dedicated technical support for process optimization. Contact our sales team for detailed specifications and volume quotations.



