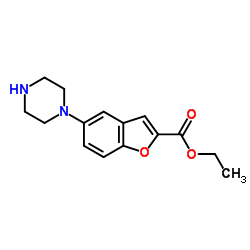
Ethyl 5-(piperazin-1-yl)benzofuran-2-carboxylate
- CAS No.163521-20-8
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate designed for Vilazodone synthesis. Offers excellent stability and assay compliance for large-scale production.
Request Bulk PricingProduct Technical Details
Product Overview
Ethyl 5-(piperazin-1-yl)benzofuran-2-carboxylate is a specialized organic compound serving as a critical building block in the pharmaceutical industry. With the CAS registry number 163521-20-8, this chemical entity is primarily utilized as a key intermediate in the multi-step synthesis of Vilazodone, a recognized therapeutic agent. Our manufacturing process ensures that each batch meets rigorous international standards for purity and consistency, making it an ideal choice for pharmaceutical research and development teams as well as large-scale commercial production facilities.
The molecular structure combines a benzofuran core with a piperazine moiety, providing unique reactivity profiles essential for downstream chemical transformations. We understand the importance of supply chain reliability in the pharmaceutical sector. Therefore, we maintain robust inventory levels and adhere to strict quality management systems to guarantee uninterrupted supply for our global partners.
Technical Specifications
Quality control is paramount in the production of pharmaceutical intermediates. Our facility employs advanced analytical techniques, including High-Performance Liquid Chromatography (HPLC), to verify the identity and purity of every lot. The following table outlines the typical physical and chemical properties associated with this product:
| Parameter | Specification |
|---|---|
| CAS Number | 163521-20-8 |
| Molecular Formula | C15H18N2O3 |
| Molecular Weight | 274.315 g/mol |
| Appearance | White to light yellow powder |
| Assay (HPLC) | ≥99.0% |
| Loss on Drying | ≤0.5% |
| Residue on Ignition | ≤0.2% |
| Individual Impurity | ≤0.2% |
| Total Impurities | ≤1.0% |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 429.0±35.0 °C at 760 mmHg |
Industrial Applications
This compound is predominantly employed as a precursor in the synthesis of complex pharmaceutical APIs. Its primary application lies in the manufacturing pathway of Vilazodone, where it acts as a crucial coupling component. The high assay purity ensures minimal side reactions during subsequent synthesis steps, thereby improving overall yield and reducing purification costs for the final active pharmaceutical ingredient. Beyond its primary use, this benzofuran derivative may also serve as a valuable reference standard or research chemical in medicinal chemistry programs focused on neurological therapeutics.
Our R&D team continuously optimizes the synthesis route to enhance environmental sustainability and cost-effectiveness. By minimizing waste and maximizing atom economy, we provide a greener alternative for clients seeking to align their supply chain with modern environmental, social, and governance (ESG) goals.
Quality Assurance and Storage
We are committed to delivering products that exceed industry expectations. Each shipment is accompanied by a comprehensive Certificate of Analysis (COA) detailing all test results against specified parameters. Our quality assurance protocol includes raw material inspection, in-process control, and final product verification.
- Stored in a cool, ventilated warehouse away from direct sunlight.
- Keep containers tightly closed when not in use to prevent moisture absorption.
- Handle with appropriate personal protective equipment (PPE) according to safety data sheets.
- Compatible with standard chemical shipping regulations for non-hazardous organic solids.
Packaging is available in 25 kg drums as standard, with customization options to suit specific logistical requirements. Whether you require small quantities for clinical trials or bulk volumes for commercial manufacturing, our logistics team ensures safe and timely delivery worldwide. Contact our sales department today to discuss your specific technical requirements and request a competitive quotation.



