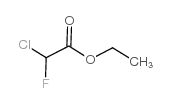
Ethyl Chlorofluoroacetate
- CAS No.401-56-9
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity ethyl chlorofluoroacetate (CAS 401-56-9), a versatile fluorinated intermediate for pharmaceutical and agrochemical R&D.
Request Bulk PricingProduct Technical Details
Product Overview
Ethyl chlorofluoroacetate (CAS 401-56-9) is a colorless, high-reactivity fluorinated ester widely employed as a key building block in advanced organic synthesis. With the molecular formula C4H6ClFO2 and a molecular weight of 140.54 g/mol, this compound serves as a critical intermediate in the development of fluorine-containing active pharmaceutical ingredients (APIs), novel agrochemicals, and specialty materials. Its unique combination of chlorine and fluorine substituents on the alpha-carbon enables selective transformations under controlled conditions, making it invaluable in modern medicinal chemistry workflows.
Specifications
| Molecular Formula | C4H6ClFO2 |
|---|---|
| Molecular Weight | 140.54 g/mol |
| Appearance | Colorless liquid |
| Density | 1.212 g/mL at 25 °C (lit.) |
| Boiling Point | 133 °C (lit.) |
| Flash Point | 125 °F |
| Refractive Index | n20/D 1.396 (lit.) |
| Vapor Pressure | 10.4 mmHg at 25 °C |
Industrial Applications
As a specialized fluorochemical intermediate, ethyl chlorofluoroacetate is primarily utilized in research and industrial-scale synthesis where precise introduction of fluorine atoms is required. Key application areas include:
- Pharmaceutical R&D: Serving as a precursor for fluorinated analogs in drug discovery programs targeting metabolic stability and bioavailability enhancement.
- Agrochemical Innovation: Enabling the construction of novel herbicidal and insecticidal scaffolds with improved environmental profiles.
- Advanced Materials: Contributing to the synthesis of fluoropolymers and functionalized monomers with tailored surface properties.
Manufacturers leverage this compound within tightly controlled fluorination platforms that ensure consistent quality, traceability, and compliance with international regulatory standards. Due to its reactivity, it must be handled under inert atmosphere and stored away from moisture and strong nucleophiles.



