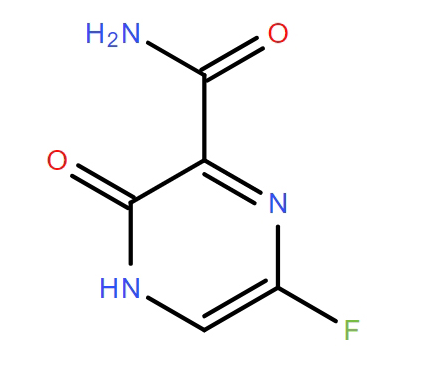
Favipiravir
- CAS No.259793-96-9
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Favipiravir intermediate available for pharmaceutical research and synthesis. Reliable supply with comprehensive COA support.
Request Bulk PricingProduct Technical Details
Product Overview
Favipiravir, chemically identified as 6-Fluoro-3-hydroxy-2-pyrazinecarboxamide, represents a significant compound in the realm of antiviral pharmaceutical research. As a pyrazinecarboxamide derivative, this substance functions as a selective inhibitor targeting viral RNA-dependent RNA polymerase. Our organization specializes in the supply of high-quality non-GMP intermediates and active pharmaceutical ingredients designed for extensive laboratory investigation and further chemical synthesis. The compound exhibits notable stability and reactivity profiles suitable for complex organic synthesis pathways.
Originally developed for stockpiling against influenza pandemics, the chemical structure of Favipiravir has garnered attention for its broad-spectrum potential against various RNA viruses. Our manufacturing processes ensure that each batch meets stringent purity requirements, facilitating reliable results in downstream applications. We maintain a robust supply chain to support global pharmaceutical factories and research institutions engaged in the development of novel therapeutic agents.
Technical Specifications
| Parameter | Specification |
|---|---|
| CAS Number | 259793-96-9 |
| Chemical Name | 6-Fluoro-3-hydroxy-2-pyrazinecarboxamide |
| Molecular Formula | C5H4FN3O2 |
| Molecular Weight | 157.104 g/mol |
| Purity | ≥98% |
| Appearance | White Powder |
| Solubility | Soluble in DMSO |
| Shelf Life | ≥ 2 Years |
Industrial Applications
This chemical intermediate is primarily utilized in the pharmaceutical sector for the synthesis of antiviral agents. Researchers employ this material to study polymerase inhibition mechanisms and to develop new formulations targeting RNA virus families including influenza, flaviviruses, and arenaviruses. The high purity grade ensures minimal interference during analytical procedures and reaction kinetics studies.
- Pharmaceutical API synthesis and process development
- Antiviral research and biological assay screening
- Reference standard preparation for quality control
- Organic chemistry educational and laboratory use
Quality Assurance and Manufacturing
Ningbo Inno Pharmchem Co., Ltd. operates advanced manufacturing bases equipped with flexible chemical synthesis systems ranging from 500mL to 3000L reactors. This infrastructure allows us to scale production efficiently while maintaining strict quality control protocols. Every shipment is accompanied by a Certificate of Analysis (COA) to verify identity and purity. We adhere to safe handling guidelines, recommending avoidance of skin contact and dust formation during usage. Our commitment to excellence ensures that clients receive consistent, high-performance materials for their critical research projects.
For bulk orders and custom synthesis requirements, our technical team provides comprehensive support to optimize your supply chain. We understand the urgency of pharmaceutical development and strive to deliver timely solutions without compromising on chemical integrity. Partner with us for reliable access to essential antiviral intermediates.
Storage and Handling
To maintain optimal stability, this product should be stored in a cool, dry place away from direct sunlight and moisture. Containers must be kept tightly closed when not in use. Personnel should utilize appropriate personal protective equipment including gloves and safety goggles during handling. Adequate ventilation is required to prevent the accumulation of airborne particles. Following these guidelines ensures safety and preserves the chemical properties for long-term use.



