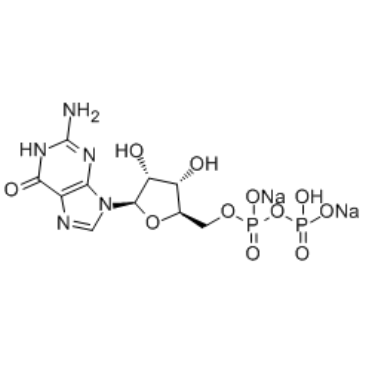
Guanosine-5'-diphosphate Disodium Salt
- CAS No.7415-69-2
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity Guanosine-5'-diphosphate disodium salt for pharmaceutical synthesis. Reliable bulk supply with comprehensive COA.
Request Bulk PricingProduct Technical Details
Product Overview
Guanosine-5'-diphosphate disodium salt is a critical nucleotide derivative widely utilized in advanced biochemical research and pharmaceutical manufacturing. As a key intermediate, this compound plays an essential role in the synthesis of complex glycoconjugates and enzyme-mediated reactions. Our facility produces this material under strict quality control standards to ensure consistent performance in sensitive applications. The disodium salt form enhances solubility and stability, making it ideal for various industrial processes requiring high reliability.
Key Specifications
| Parameter | Value |
|---|---|
| CAS Number | 7415-69-2 |
| Molecular Formula | C10H13N5Na2O11P2 |
| Molecular Weight | 487.16 g/mol |
| Appearance | White to off-white crystalline powder |
| Assay (HPLC) | ≥98.0% |
| Loss on Drying | ≤15% |
| Storage Condition | Cool, ventilated place |
Industrial Applications
This chemical serves as a fundamental building block in the production of pharmaceutical intermediates. It is extensively used in the development of nucleotide sugars which are vital for glycosylation processes in biotechnology. Researchers utilize this compound for enzyme kinetic studies, particularly involving glycosyltransferases. The high purity grade ensures minimal interference in downstream synthesis routes, supporting the creation of high-value therapeutic agents. Our manufacturing process is optimized to meet the rigorous demands of the global pharmaceutical supply chain.
- Pharmaceutical intermediate synthesis
- Biochemical research and enzyme assays
- Production of nucleotide sugars
- Quality control reference standard
- Industrial scale glycosylation reactions
Quality Assurance and Handling
We adhere to comprehensive quality management systems to guarantee product integrity. Every batch undergoes rigorous testing using high-performance liquid chromatography to verify purity levels. Certificates of Analysis (COA) are provided with each shipment to confirm compliance with specified parameters. Proper handling procedures are recommended to maintain stability. The product should be stored in a cool, ventilated environment away from direct sunlight and moisture. Packaging is designed to protect the material during international transit, ensuring it arrives in optimal condition.
Packaging and Logistics
Standard packaging includes 25 kg drums, suitable for large-scale industrial use. Customized packaging solutions are available upon request to meet specific client requirements. We offer global shipping capabilities with secure logistics partners to ensure timely delivery. Our team provides full documentation support for customs clearance and regulatory compliance. For bulk orders, competitive pricing structures are available to support long-term partnerships. Contact our sales team for detailed quotations and technical support regarding synthesis routes.



