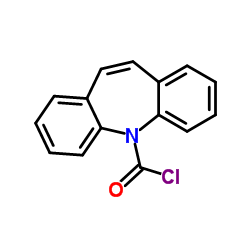
Iminostilbene N-Carbonyl Chloride
- CAS No.33948-22-0
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity Iminostilbene N-Carbonyl Chloride intermediate for pharmaceutical synthesis. Reliable supply with comprehensive COA and global shipping capabilities.
Request Bulk PricingProduct Technical Details
Product Overview
Iminostilbene N-Carbonyl Chloride is a highly specialized organic chemical intermediate primarily utilized within the pharmaceutical manufacturing sector. This compound serves as a crucial building block in the synthesis of complex therapeutic agents, specifically acting as a key precursor in the production of anticonvulsant medications. Our facility ensures that every batch meets rigorous international standards for chemical purity and consistency, making it an ideal choice for large-scale industrial applications.
As an acid chloride derivative, this compound exhibits specific reactivity profiles that require careful handling and precise storage conditions. We provide comprehensive technical support to ensure safe integration into your existing synthesis routes. Our commitment to quality assurance means that clients receive material that facilitates efficient downstream processing with minimal impurity carryover.
Technical Specifications
| Parameter | Specification |
|---|---|
| Chemical Name | Iminostilbene N-Carbonyl Chloride |
| CAS Number | 33948-22-0 |
| Molecular Formula | C15H10ClNO |
| Molecular Weight | 255.699 g/mol |
| Appearance | White to yellow powder |
| Assay | ≥95.0% |
| Melting Point | 149-153 °C |
| Boiling Point | 413.7±38.0 °C |
| Density | 1.3±0.1 g/cm3 |
| Flash Point | 204.0±26.8 °C |
Industrial Applications
The primary application of this intermediate lies in the pharmaceutical industry, where it functions as an essential synthon for the manufacture of Carbamazepine and related derivatives. High purity levels are critical in this context to ensure the safety and efficacy of the final therapeutic product. Our manufacturing process is optimized to minimize side products that could complicate subsequent reaction steps.
- Pharmaceutical API synthesis precursor
- Organic synthesis building block
- Research and development reagent
- Custom chemical manufacturing feedstock
Quality Assurance and Storage
We maintain a strict quality control protocol that includes advanced analytical testing such as HPLC and GC-MS to verify composition and purity levels. Each shipment is accompanied by a Certificate of Analysis (COA) detailing batch-specific data. Proper storage is essential to maintain stability; the product should be kept in a cool, ventilated environment away from moisture and incompatible materials.
Packaging options include standard 25 kg drums, with flexibility for custom packaging solutions based on client logistical requirements. We adhere to global safety regulations for the transport of chemical materials, ensuring secure delivery to our partners worldwide. Our team is available to provide safety data sheets and handling guidelines upon request.



