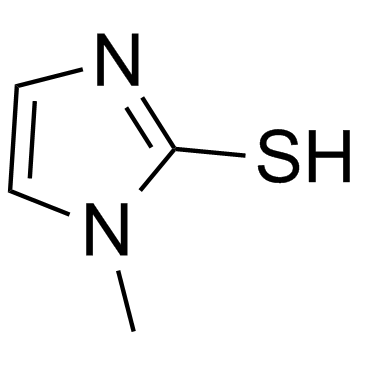
Methimazole
- CAS No.60-56-0
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Methimazole (CAS 60-56-0) supplied for pharmaceutical synthesis. Compliant with strict quality standards for antithyroid intermediate production.
Request Bulk PricingProduct Technical Details
Product Overview
Methimazole, chemically known as 1-methyl-1H-imidazole-2-thiol, is a critical heterocyclic compound widely utilized within the pharmaceutical industry. As a key building block, it serves primarily as an active pharmaceutical ingredient (API) and as a vital intermediate in the synthesis of carbimazole. Our facility specializes in the production of this compound, adhering to rigorous international quality standards to support global pharmaceutical manufacturing needs.
This compound is characterized by its distinct imidazole structure containing a thiol group, which contributes to its specific reactivity profiles essential for downstream medicinal chemistry. We ensure that every batch undergoes comprehensive analytical testing to verify identity, potency, and impurity profiles, guaranteeing reliability for large-scale synthesis operations.
Technical Specifications
Our manufacturing process is designed to achieve high consistency and purity levels. The physical and chemical properties are tightly controlled to meet the demands of sensitive organic synthesis pathways. Below are the standard specifications for our commercial grade material.
| Parameter | Specification |
|---|---|
| CAS Number | 60-56-0 |
| Molecular Formula | C4H6N2S |
| Molecular Weight | 114.17 g/mol |
| Appearance | White to off-white crystalline powder |
| Assay (Purity) | ≥99.0% |
| Melting Point | 144-147 °C |
| Loss on Drying | ≤0.5% |
| Residue on Ignition | ≤0.1% |
| Heavy Metals | Comply with limits |
Industrial Applications
The primary application of this material lies within the pharmaceutical sector, specifically for the development of antithyroid medications. It functions as a crucial precursor in the manufacturing chain for therapies aimed at regulating thyroid function. Due to its specific chemical structure, it is also utilized in research and development settings for exploring new heterocyclic derivatives.
For process chemists, maintaining high industrial purity is essential to minimize side reactions during subsequent synthesis steps. Our product is optimized to provide a reliable synthesis route, reducing the need for extensive purification downstream. This efficiency supports cost-effective manufacturing processes for finished dosage forms.
Quality Assurance and Handling
Quality control is paramount in our production facility. Each lot is accompanied by a Certificate of Analysis (COA) detailing test results against established specifications. We employ advanced chromatographic and spectroscopic methods to confirm the absence of organic volatile impurities and ensure selenium levels remain within safe thresholds.
- Strict compliance with Good Manufacturing Practices (GMP) guidelines.
- Comprehensive stability testing to ensure shelf-life integrity.
- Secure packaging to prevent moisture absorption and degradation.
Packaging and Storage
To maintain product integrity during transit and storage, Methimazole is packaged in 25kg cardboard drums or according to specific customer requirements. The material should be stored in a cool, dry, and well-closed container. It is imperative to keep the substance away from strong light, heat, and moisture to prevent chemical instability. Proper handling procedures ensure that the physical properties remain consistent from the point of manufacture to the point of use.



